Steps to Better Specimen Collection and Transport

From patient to pathologist, preparing tissue specimens for histological examination requires care, skill and sound procedures. This guide provides practical advice on best practice techniques and simple ways to avoid common errors.
Tips for better specimen collection and transport are highlighted in this guide. We hope each step provides a valuable reminder of good histology practice and helps with troubleshooting when unacceptable results do occur.
Want to see all 101 Steps to Better Histology?
Download 101 Steps to Better Histology now!
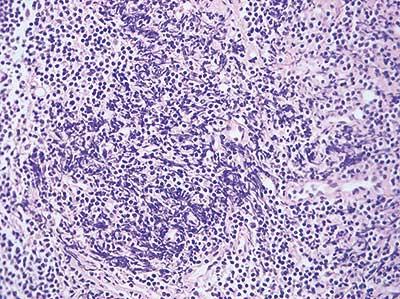
Avoid Mechanical Trauma
Tissue is removed gently to avoid trauma to the specimen caused by crushing or tearing. This applies both during surgery and during any further dissection that may be required of a fresh specimen.
Specimen is damaged before fixation by crushing or tearing during removal.
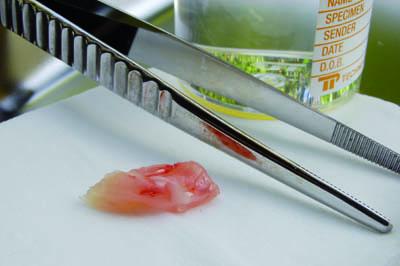
Prevent Specimen Drying
Specimen is not allowed to dry out prior to fixation. If immediate fixation is not practicable, gauze moistened with saline can be used to prevent this.
Specimen is left on absorbent surface for some time prior to fixation.
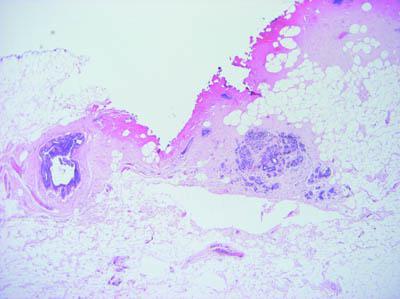
Avoid Heat Damage
As far as possible avoid local heat damage to specimens (some damage by cautery may be unavoidable).
Any unnecessary local heat applied to tissue will cause damage. Fresh tissue is particularly susceptible.
Avoid Chemical Damage
Avoid contaminating fresh specimens with foreign chemicals or substances such as disinfectants.
The surface of unfixed tissue can be easily penetrated and damaged by foreign reagents or substances.
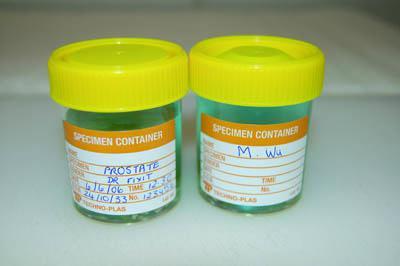
Label Specimens Properly
Each specimen should be properly identified, and all details recorded as soon as possible.
Recording of specimen details is delayed and the information provided is incomplete.
Ensure Prompt Fixation
Fixation is always carried out promptly. If it is necessary that a specimen remains unfixed for a short period of time, it should be refrigerated at 4 °C.
Fixation is delayed (degeneration of tissue elements commences as soon as the specimen is deprived of a blood supply).

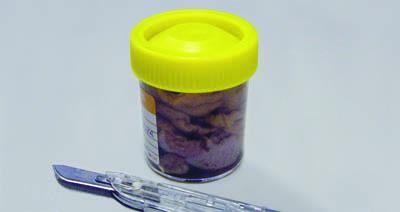
Use Sufficient Fixative and a Suitable Container
An adequate volume of fixative (ratio of at least 20:1) is used in a container of an appropriate size. This avoids distortion of the fresh specimen and ensures good quality fixation.
Specimens are sometimes squashed into a small container with insufficient fixative to cover the specimen surface.
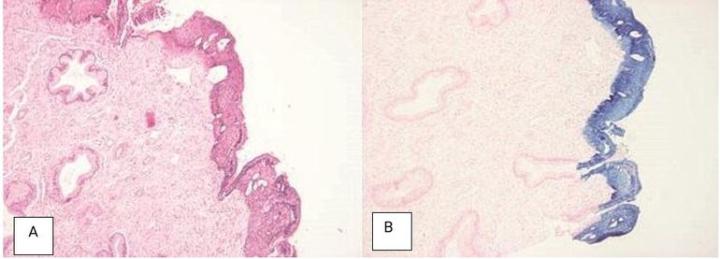
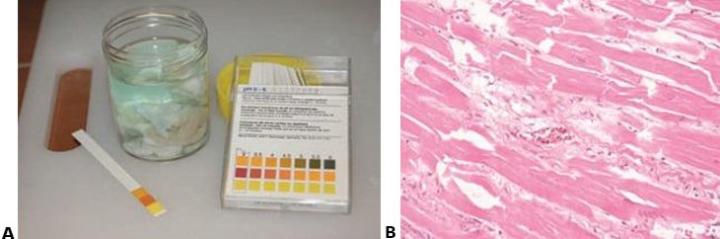
Check Fixative pH
The fixative is of high quality and at the optimal pH.
The fixative is of poor quality and unknown pH. If formalin is used at acid pH it rapidly produces “formalin pigment” by reaction with hemoglobin. Near neutral solutions will still produce the pigment but much more slowly. In good histological preparations, formalin pigment should be removed prior to staining.
Expedite Large Specimen Fixation
The specimen dimensions allow rapid penetration of the fixative. Large specimens should be rapidly transported to the lab to allow grossing (tissue slices can be prepared to allow proper fixation to occur).
Large specimens are left in fixative for an extended time prior to grossing. The center of the specimen may remain unfixed and the tissue can become markedly distorted.

Avoid Unnecessary Delays
No unnecessary delays – specimen reaches lab in minimum time.
Specimens are sometimes delayed, specimen transport has a low priority and is not well organized.

Handle Specimens Gently
Specimens handled gently – fragile specimens remain intact.
Specimens handled roughly – delicate friable specimens can be damaged.

About the presenter

Geoffrey Rolls is a Histology Consultant with decades of experience in the field. He is a former Senior Lecturer in histopathology in the Department of Laboratory Medicine, RMIT University in Melbourne, Australia.
Related Content
Leica Biosystems Knowledge Pathway content is subject to the Leica Biosystems website terms of use, available at: Legal Notice. The content, including webinars, training presentations and related materials is intended to provide general information regarding particular subjects of interest to health care professionals and is not intended to be, and should not be construed as, medical, regulatory or legal advice. The views and opinions expressed in any third-party content reflect the personal views and opinions of the speaker(s)/author(s) and do not necessarily represent or reflect the views or opinions of Leica Biosystems, its employees or agents. Any links contained in the content which provides access to third party resources or content is provided for convenience only.
For the use of any product, the applicable product documentation, including information guides, inserts and operation manuals should be consulted.
Copyright © 2026 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.