The Benefit Of Multiple Tissue Processing Runs

Tissue processing is one of the most critical elements of obtaining quality and timely results in the histology process. Tissue processing in the laboratory prepares a specimen by infiltrating the tissue with paraffin wax to cut into thin slices in preparation for staining. Fixatives, processing protocols, tissue type, and size are critical factors for optimal tissue processing results.1 According to Frieda Carson, the biggest problem when processing tissue is processing both biopsy and large [routine] tissue specimens in the same processing run.2 Tissue can become over-processed – that is, the tissue may appear to be cooked, brittle, crisp, dry, or powdery, or under-processed and cannot be sectioned. Biopsy and larger sample tissues should be processed separately using a protocol specific to the specimen.3
Method
This study, conducted by the Leica Biosystems Content and Evidence Team, occurred at a small independent laboratory. The laboratory was interested in a complete review of their current processes and recommendations for quality and turn-around time improvements. As a result of the initial observations, the team focused on the laboratory’s tissue processing practices.
Results
Discussion
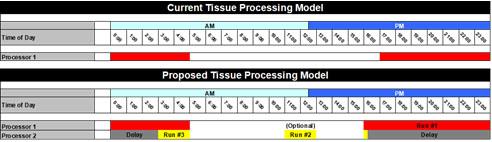
The Leica Biosystems Content and Evidence Team examined the laboratory’s current tissue processing schedule. The laboratory uses a single tissue processor with one retort for one overnight batch run for all specimen types, including biopsies and routine tissue. Highly variable sizes in biopsy tissue versus routine tissue, and even fatty tissue, require significant time changes in the processing reagents. Combining tissue types into one overnight batch run can result in over-processing biopsies while not fully processing larger routine tissue. Tissue that is not initially processed sufficiently is pulled out of the daily workload and reprocessed on the next overnight processing run, delaying turn-around time on those cases by more than 24 hours.
As a result of this assessment, the recommendation is to separate processing schedules to accommodate the various tissue types and sizes:
- Run #1 overnight for 12 hours for the routine specimens
- Run #2 (optional) during the day for 2 hours for biopsies
- Run #3 a delayed run for biopsies received during the late afternoon
To maintain the current workflow and the overlap of runs (that is, the delayed biopsy and 12-hour run), this processing model could only be accomplished by moving from 1 tissue processor to 2 tissue processors or from a single retort processor to a dual retort processor.
Conclusion
Ultimately, to obtain the desired quality processed tissue samples, separate processing runs may be required appropriate to tissue size. The addition of a small biopsy run during the day, a delayed run for biopsies, and an overnight extended run for routine tissue can increase throughput and reduce turn-around time for biopsies.
Projections and Realized Results are specific to the institution where they were obtained and may not reflect the results achievable at other institutions.
About the presenter

Ashley Troutman has been involved in Laboratory Medicine for more than 20 years in clinical, research and administrative capacities. He has worked in facilities of all sizes, from small community hospitals and private labs to large academic medical centers and corporate reference labs. He has extensive experience in laboratory science and management, specifically in anatomic pathology and immunohistochemistry. He has managed routine histology operations and has been part of the team to aid researchers in designing experiments using histologic techniques. These roles have allowed Ashley to lead work process implementation teams that saw success in scientific innovation as well as improving laboratory efficiency through areas of waste/cost reduction, process improvement and safety.
References
- Survarna, S. Kim, Layton, Christopher and Bancroft, John D.; Bancroft’s Theory and Practice of Histological Techniques; 7th edition; 2013
- Carson, Frieda L.; Histologic Fixation and Sample Processing;
- Bancroft’s Theory and Practice of Histological Techniques: Expert Consult
Related Content
Leica Biosystems Knowledge Pathway content is subject to the Leica Biosystems website terms of use, available at: Legal Notice. The content, including webinars, training presentations and related materials is intended to provide general information regarding particular subjects of interest to health care professionals and is not intended to be, and should not be construed as, medical, regulatory or legal advice. The views and opinions expressed in any third-party content reflect the personal views and opinions of the speaker(s)/author(s) and do not necessarily represent or reflect the views or opinions of Leica Biosystems, its employees or agents. Any links contained in the content which provides access to third party resources or content is provided for convenience only.
For the use of any product, the applicable product documentation, including information guides, inserts and operation manuals should be consulted.
Copyright © 2026 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.