Potential to Improve Patient Care Through Identifying Inefficiencies
The Leica Biosystems Process and Solutions Optimization team partnered with a private teaching hospital. Hospital Administration was interested in several areas pertaining to the breast care program including:
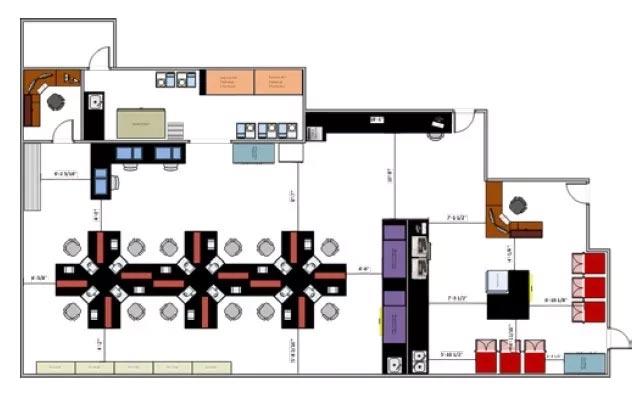
Initial Data
- Recommendations and current evidence to inform and encourage improved surgical timeliness to care and adjuvant therapy after initial biopsy
- Recommendations relative to process and efficiency improvements for breast needle localizations followed by surgeries
- Recommendations for reduction in outmigration
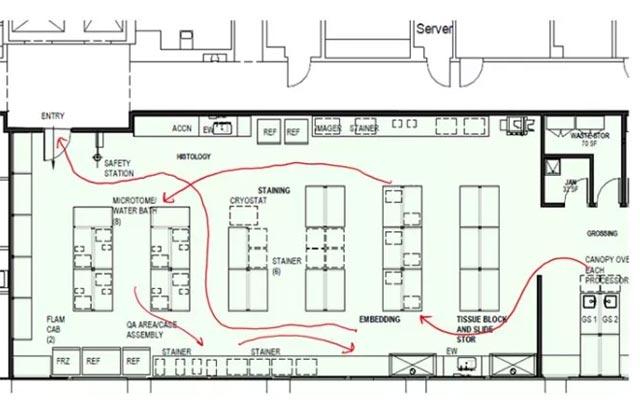
The Leica Biosystems team conducted a three-day on site visit to the breast center and found several opportunities for improvement.

Recommendations
Potential to improve efficiencies from biopsy to surgery
Use of needle localizations was a cumbersome process and was identified to cause delay in surgery. Earlier start times for needle localizations were instituted. The team should explore, pilot, and record any efficiencies from the move of the use of needle localizations to the magnetic seed to see if there is improvement in surgical timeliness.
Potential to improve efficiencies from surgery to adjuvant therapy
Oncotype Dx testing: The test is intended for use in all newly diagnosed patients with early-stage (stage I, II or IIIa), breast cancer who have node-negative or node-positive (1-3), estrogen receptor-positive (ER+), HER2 -negative disease, and with DCIS for determination of appropriate therapy. The test can be run on a post-lumpectomy tumor sample or on a core biopsy. Oncotype Dx should be ordered by the breast surgeon, so it does not delay further treatment. Turnaround time for the test is approximately two weeks.
Program Promotion
The Leica Biosystems team recommends this facility should display banners or information regarding the CoC accreditation, NAPBC accreditation and Center of Excellence awards.
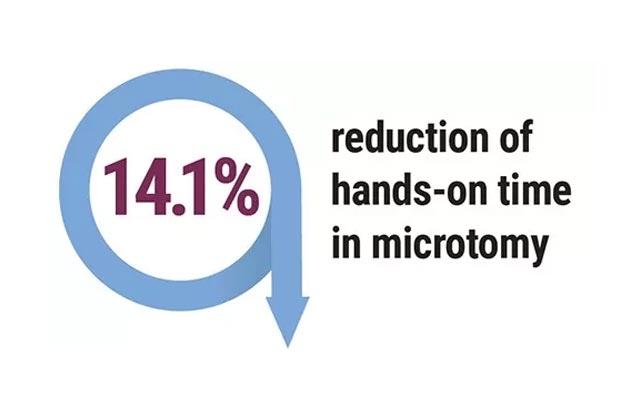
Projected Outcomes*
Projected Outcomes include:
- Decrease in the number of days from biopsy to initial surgery – currently at 31 days.
- Decrease in the number of days from biopsy to adjuvant therapy – currently at 68–77 days.
- Reduction of the outmigration percentage – currently at 19.3%.
Projections and Realized Results are specific to the institution where they were obtained and may not reflect the results achievable at other institutions.
Want to see how the Leica Process and Solutions Optimization team can help improve your lab's workflow?
Related Content
Die Inhalte des Knowledge Pathway von Leica Biosystems unterliegen den Nutzungsbedingungen der Website von Leica Biosystems, die hier eingesehen werden können: Rechtlicher Hinweis. Der Inhalt, einschließlich der Webinare, Schulungspräsentationen und ähnlicher Materialien, soll allgemeine Informationen zu bestimmten Themen liefern, die für medizinische Fachkräfte von Interesse sind. Er soll explizit nicht der medizinischen, behördlichen oder rechtlichen Beratung dienen und kann diese auch nicht ersetzen. Die Ansichten und Meinungen, die in Inhalten Dritter zum Ausdruck gebracht werden, spiegeln die persönlichen Auffassungen der Sprecher/Autoren wider und decken sich nicht notwendigerweise mit denen von Leica Biosystems, seinen Mitarbeitern oder Vertretern. Jegliche in den Inhalten enthaltene Links, die auf Quellen oder Inhalte Dritter verweisen, werden lediglich aus Gründen Ihrer Annehmlichkeit zur Verfügung gestellt.
Vor dem Gebrauch sollten die Produktinformationen, Beilagen und Bedienungsanleitungen der jeweiligen Medikamente und Geräte konsultiert werden.
Copyright © 2026 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.