
The Basic Chemistry of Hematoxylin

Staining protocols utilizing hematoxylin are the most commonly used of the routine staining procedures performed in the histology laboratory. In addition, hematoxylin-based protocols are among the oldest in the laboratory as some of the formulations of hematoxylin-based solutions date back over one hundred years. The value of hematoxylin lies in the capacity of aluminum hematoxylin solutions to stain the chromatin of cell nuclei. Although the recent advances in cell biology and molecular pathology have resulted in significant improvements in the practice of diagnostic pathology, the changes in chromatin patterns together with alterations of nuclear size and shape that are revealed with hematoxylin stains remain key diagnostic markers in the evaluation of pathological changes. In spite of the importance and relevance of hematoxylin in diagnostic pathology, the chemistry of hematoxylin remains poorly understood. The following article is a review of the current state of understanding of the chemistry of aluminum-based hematoxylin stains and the mechanism by which these stains act.
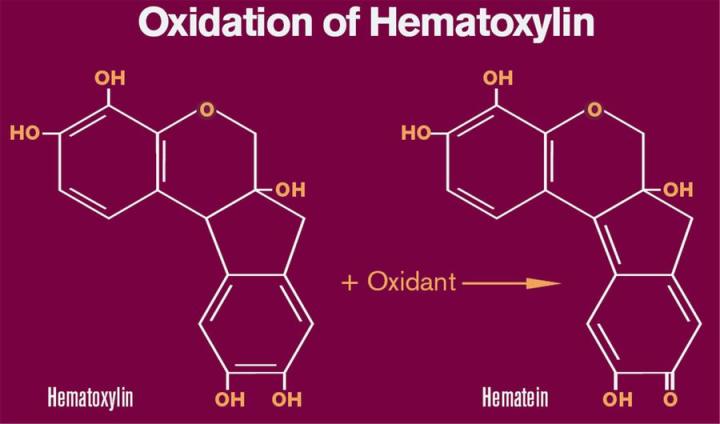
Oxidation and Formation of Hematein
Hematoxylin is a natural product extracted from the heartwood of the logwood tree (Haematoxylum campechianum). Hematoxylin is relatively colorless and without further modifications has little or no value as a biological stain. The use of the word “hematoxylin” to describe a staining solution is somewhat misleading but continues to be used as a term of convenience. To produce a functional dye, hematoxylin is oxidized to hematein and subsequently is bound to one of several metal ions including aluminum (Al+3), iron (Fe+3) and chromium (Cr+3). A metallic ion bound to a dye that is involved in the binding of the dye to tissue is referred to as a mordant. This article is concerned specifically with the chemistry of aluminum bound hematoxylin as a nuclear stain.
Conversion of hematoxylin to hematein may be accomplished by the action of a number of agents. Several older formulations such as Delafield’s and Ehrlich’s rely on atmospheric oxygen for oxidation. In these formulations, hematoxylin and aluminum salts are stored in loosely capped or cotton plugged bottles to facilitate oxidation. The process of bubbling air through hematoxylin solutions also has been used to speed up the oxidation process. Due to the extended period of time (4 - 10 weeks) necessary to “naturally ripen” or oxidize hematoxylin under these conditions, these procedures are rarely used today. Currently most formulations incorporate a chemical oxidant such as sodium iodate that rapidly converts hematoxylin to hematein. Concentrations of sodium iodate typically are based upon the amount of hematoxylin and usually range from 0.10 to 0.20 grams of sodium iodate per gram of hematoxylin. Mercuric oxide was used as the oxidant of choice in Harris formations for many years but is no longer used due to environmental issues concerning mercury. Today, mercuric oxide is replaced with sodium iodate in modified Harris formulations.
In contrast to hematoxylin which has been well characterized structurally, the molecular structure of hematein has been debated in recent years. The traditional structure of hematein which includes a quinoid ring is depicted in Figure 1. Regardless of the exact structure of hematein, we can assume that hematein is structurally similar to hematoxylin based upon NMR analysis and similarities in molecular weight. The change in structure results in the conversion of the relatively colorless hematoxylin to the reddish/brown hematein. In addition, the oxidation to hematein is necessary in order to bind metal ions such as aluminum.
Dye Lake Formation
A number of different aluminum salts are used as a source of Al+3.These include aluminum ammonium sulfate [AlNH4(SO4)2], aluminum sulfate [Al2(SO4)3] and aluminum potassium sulfate [AlK(SO4)2]. Mayer’s and Harris formulations typically contain AlNH4(SO4)2 or AlK(SO4)2, while Gill formations contain Al2(SO4)3. There is in fact probably little difference among the aluminum salts other than the amount of Al+3 that each can contribute to the solution. AlNH4(SO4)2 and AlK(SO4)2 have limited solubility in aqueous solutions and under most conditions do not completely ionize to produce free Al+3.
While it is generally accepted that hematein forms a complex or “lake” with Al+3, the nature or structure of this complex has yet to be fully elucidated. The ratio of Al+3 to hematein molecules as well as the overall charge of the hematein-Al+3 complex has been a subject of debate over the past 50 years. Most evidence, supports the concept that hematein-Al+3 complexes are positively charged or “cationic” at most staining conditions.
In addition to the oxidants and aluminum salts described above, hematoxylin solutions often contain other additives or agents. Modifications to the solvent are made by the addition of polyhydroxy alcohols such as glycerin or more commonly ethylene glycol. Initially it was proposed that the polyhydroxy alcohols acted as stabilizers to reduce the over oxidation of hematein. In fact, these additives primarily act to increase the solubility of hematein and the hematein-Al+3 complexes within the solution. Although hematoxylin is relatively soluble in aqueous solutions, hematein and hematein-Al+3 are only sparingly soluble in water. Hematein and hematein-Al+3 may precipitate and form a sediment or a metallic sheen on the surface of the solution. The inclusion of ethylene glycol in the solution reduces the precipitation or deposition of these products and thus increase the stability or shelf life of the hematoxylin solution.
Various acids are sometimes added to hematoxylin solutions to reduce the pH. Acetic acid is added to Gill type hematoxylins while citric acid is found in Mayers. The addition of acids to hematoxylin solutions increases the selectivity of the stain for nuclei by reducing nonspecific background staining. The effects of reduced pH on staining is discussed in greater detail in the section “Mechanism(s) of Staining”.
Mechanism(s) of Staining
A number of varied theories have been proposed to account for the staining of nuclei by hematein-Al complexes. Nuclear molecules that have been proposed to bind hematein-Al complexes include basic (positively charged) proteins such as histones as well as deoxyribonucleic acid (DNA). There is currently little evidence to support the suggestion that basic nuclear proteins bind hematein complexes. Evidence is more consistent with the theory that the hematein-Al+3 complexes bind DNA. Evidence to support this theory includes studies utilizing DNase enzymes. These studies have demonstrated that digestion of nuclear DNA prevents staining of all nuclei with hematein/Al solutions.
The attraction or binding of hematein-Al+3 to DNA is likely due to the electrostatic attraction of the cationic hematein-Al+3 complexes for the phosphate groups as these groups carry a negative charge at staining conditions. This theory is also consistent with the observation that other anionic sites such as those found in mucins and proteoglycans may create background staining by binding hematein-Al+3. In fact, background staining may be reduced by lowering the pH of the staining of hematein-Al+3 solutions. A reduction of pH can be expected to protonate (addition of H+) many of these sites such that they become neutral. The fact that these substances at a neutral charge no longer bind the positively charged hematein-Al+3 complexes further substantiates the role of electrostatic forces as mediators of hematein-Al+3 binding.
It has been suggested that simple electrostatic forces alone are not sufficient to account for the staining of nuclei with Al/hematein solutions. This theory is based upon the belief that the electrostatic forces mediate the initial attraction of the hematein-Al+3 complexes for DNA but the electrostatic bonds are replaced with more stable covalent or coordinate covalent bonds. The greater stability of the covalent type bonds would account for the relative permanent nature of hematein-Al+3 staining. There is, however, little direct evidence to support the role of covalent bond formation in the binding of hematein-A+3 complexes to DNA.
Figures
About the presenter

Russell Myers acquired his Ph.D. in endocrine physiology from the Medical College of Georgia. Russell Myers is responsible for design and development of reagents and consumables used in the histology laboratory.
Leica Biosystems Knowledge Pathway content is subject to the Leica Biosystems website terms of use, available at: Legal Notice. The content, including webinars, training presentations and related materials is intended to provide general information regarding particular subjects of interest to health care professionals and is not intended to be, and should not be construed as, medical, regulatory or legal advice. The views and opinions expressed in any third-party content reflect the personal views and opinions of the speaker(s)/author(s) and do not necessarily represent or reflect the views or opinions of Leica Biosystems, its employees or agents. Any links contained in the content which provides access to third party resources or content is provided for convenience only.
For the use of any product, the applicable product documentation, including information guides, inserts and operation manuals should be consulted.
Copyright © 2024 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.