
An Innovative Methodology to Measure Product Usability Histotechnicians are at the Center of our Focus

Whether it’s a new imaging device or a software product, at Leica Biosystems we want to make our products so simple to use that they naturally fit into the Histotechnician’s daily life in a Pathology lab. To do this, we follow a testing process to measure product usability during product development and design of a new product. We refer to this testing methodology as User Experience (UE). Although we study multiple users, including Pathologists, IT professionals and Lab Directors, in this article we will discuss how we measure usability specifically with Histotechnicians using, among other techniques, an innovative approach called the System Usability Scale (SUS).1
What Usability Metrics do we Measure?
When measuring the User Experience of a product during the design phase, we visit Pathology labs around the world and test prototypes with many Histotechnicians. We prepare testing scenarios that the Histotechnician will experience in their day-to-day work life.
During learning cycles we test, measure and record the following:
- System Usability Scale (SUS) for each Histotechnician after each testing session.
- Task completion rate and time to complete typical tasks for which the product will be used
- Ease of use score per the Histotechnician feedback after performing each task
- Direct verbal feedback from Histotechnicians after testing the product
What is the System Usability Scale (SUS)? SUS is a ten-item likert scale that is designed to assess overall opinions of a product (or technology) in development by answering 10 survey questions regarding a product tester’s opinion of product usability, complexity, support needed, integration ability, and confidence using the product.
SUS results are expressed as percentages with low scores being in the 55-69, average scores being 70-85, an above average scores being 85-100.
Design, Test with User, Iterate, Repeat Process
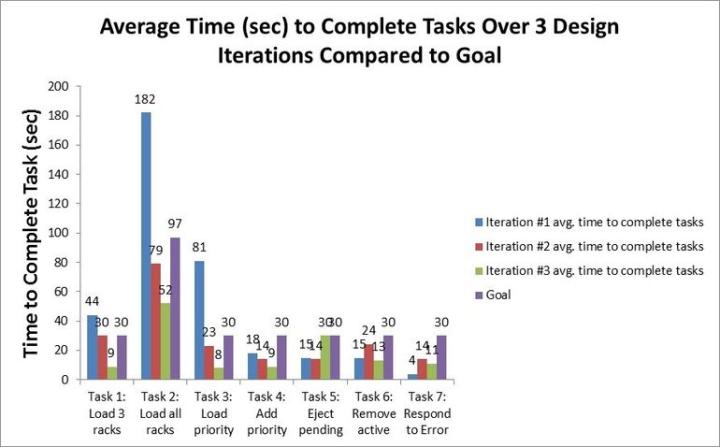
Once we have measured the usability during a learning session in a Pathology lab, we take those scores and all the rich learnings back to our Pathology Imaging headquarters and study the results in an Obeya Room (Obeya means “large room” or “war room”). In this room, we carefully study the rich user data as a team, including engineers, software developers, marketers, and product managers. We then identify which changes are needed to the product before going out for the next learning and testing session with the Histotechnician. The changes to the product are, made and then we repeat the testing and measuring process. See below for some UX DNA data for which we collected during a recent product development program.
| System Usability Score (%) | ||
| Design Iteration | Actual | Goal |
| Iteration #1 | 73% | 85% |
| Iteration #2 | 73% | 85% |
| Iteration #3 | 91% | 85% |
| Iteration #4 | 89% | 85% |
| Iteration #5 | 94% | 85% |
Table 1 (above): System Usability Scales (SUS) measurements after product testing sessions over several iterations. SUS are designed to assess overall opinions of a product (or technology) in development by answering 10 survey questions regarding a product tester’s opinion of product usability, complexity, support needed, integration ability, and confidence using the product. Results are expressed as a percent; with 40% being a poor grade and >85% being an excellent grade (as the table indicates, SUS increased from 73% to 94% over 5 product design iterations indicating excellent scores by iteration #5.) SUS results are expressed as percentages with low scores being in the 55-69, average scores being 70-85, an above average scores being 85-100.
A High Quality Product Experience for Our Customers
In order to release a product to our valued customers, Leica Biosystems is committed to achieving high scores in User Experience testing before the product’s design is completed. Often times, this may require many rounds of user testing and associated design changes before a product is ready to launch. This rigorous process is very important for our customers as it helps ensure that when a new product is introduced into the Pathology Lab it will be easy to operate and provide high value to the customer.
For more short innovation articles like this, please go to LeicaBiosystems.com/Knowledge Pathway.
Coming Soon! Look for our next article on Leica Biosystems innovations!
About the presenter

Lance Mikus earned his bachelor’s degree from UCLA. He worked as a scientist and lab manager at the UW-Madison Medical School, resulting in several publications, and went on to earn an MBA focused on Entrepreneurship and Marketing from the UW-Madison School of Business. Mr. Mikus has 20 years of commercial operations experience in the healthcare industry. He also holds a green belt in six sigma quality practices, is a certified practitioner in launch excellence and has been awarded 5 patents.
Referencias
1. Brooke, J. (1986). "SUS: a "quick and dirty" usability scale". In P. W. Jordan, B. Thomas, B. A. Weerdmeester, & A. L. McClelland (eds.). Usability Evaluation in Industry. London: Taylor and Francis.
Related Content
El contenido de Leica Biosystems Knowledge Pathway está sujeto a las condiciones de uso del sitio web de Leica Biosystems, disponibles en: Aviso legal.. El contenido, incluidos los webinars o seminarios web, los recursos de formación y los materiales relacionados, está destinado a proporcionar información general sobre temas concretos de interés para los profesionales de la salud y no está destinado a ser, ni debe interpretarse como asesoramiento médico, normativo o jurídico. Los puntos de vista y opiniones expresados en cualquier contenido de terceros reflejan los puntos de vista y opiniones personales de los ponentes/autores y no representan ni reflejan necesariamente los puntos de vista ni opiniones de Leica Biosystems, sus empleados o sus agentes. Cualquier enlace incluido en el contenido que proporcione acceso a recursos o contenido de terceros se proporciona únicamente por comodidad.
Para el uso de cualquier producto, debe consultarse la documentación correspondiente del producto, incluidas las guías de información, los prospectos y los manuales de funcionamiento.
Copyright © 2026 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.



