Optimizing Microtomy, Case Assembly and IHC Testing Through Implementation of Automated Specimen Tracking
The Leica Biosystems Content and Evidence Team has partnered with a private, multi-specialty clinic to assess the impact of implementing automated specimen tracking into their laboratory. This laboratory is most interested in increasing efficiency and patient safety throughout their processes.
During the Optimization Assessment, the Content and Evidence Team observed current processes throughout the histologic process beginning with accessioning and following through to case assembly. As with many laboratories without automated specimen tracking, paper logs or pending reports are used to track samples and users. The staff at this facility currently use LIS generated pending reports to follow cassettes and slides through their processes. Technologists refer to the reports for necessary sample information and to initial the steps that they perform to those samples.
The Leica Biosystems Content and Evidence Team also observed the current processes involved in creating slides. Slide labels are created while grossing dictation is entered into the LIS . The labels are printed from the LIS for each case. The labels are then delivered to the histology area of the laboratory in large groups. The staff spend time placing the labels onto the selected color slides and sorting them by case type and number. When cassettes are ready for microtomy, the technologists search for the matching slides and create a batch. Because there is such a large possibility of matching errors, the laboratory performs a manual block check verification with the slides before sending them to a pathologist.
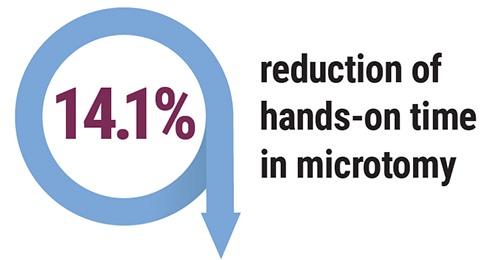
Through the implementation of an automated specimen tracking system, this laboratory could move towards eliminating the use of the LIS reports and change their labeling and slide creation process to a safer on-demand process. The resulting on-demand slide creation would be verified in the specimen tracking system to ensure that the tissue placed on the slide is from the correct cassette. This would help eliminate the chance of a mis-label during the labeling process in microtomy and reduce the safety risk to the patient. By eliminating these currently required manual steps, the average hands-on time would drop from 171.4 seconds to 147.2 seconds per block in microtomy.
Another area in which a specimen tracking system would increase efficiency is in immunohistochemistry. Since the specimen tracking system could be integrated with their IHC instrumentation, there would no longer be a need for the manual stain ordering and the assignment step. That integration could save more than 29 seconds per slide of hands-on time.
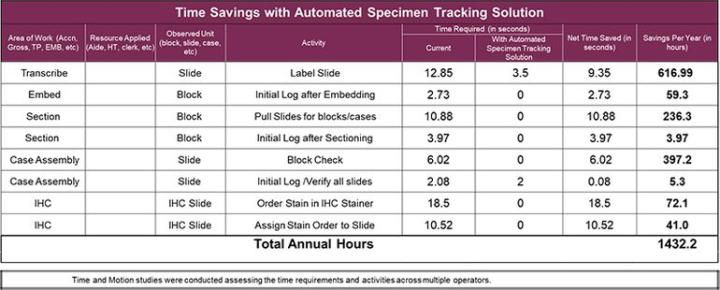
Since the specimen tracking system maintains the necessary information and tracks users throughout the process, the need to refer to or initial logs should be eliminated. The chart below shows specific steps and times savings that this laboratory could realize.
- Elimination of pending log used in 3 areas of the laboratory
- Save approximately 1432 hours/year by eliminating manual tasks
- Reduction of 14.1% hands-on time at microtomy
- Elimination of possible mis-label at grossing and microtomy
- Reduction of 29 seconds per slide in IHC
Projections and Realized Results are specific to the institution where they were obtained and may not reflect the results achievable at other institutions.
Want to see how we can help improve your lab's workflow?
Related Content
El contenido de Leica Biosystems Knowledge Pathway está sujeto a las condiciones de uso del sitio web de Leica Biosystems, disponibles en: Aviso legal.. El contenido, incluidos los webinars o seminarios web, los recursos de formación y los materiales relacionados, está destinado a proporcionar información general sobre temas concretos de interés para los profesionales de la salud y no está destinado a ser, ni debe interpretarse como asesoramiento médico, normativo o jurídico. Los puntos de vista y opiniones expresados en cualquier contenido de terceros reflejan los puntos de vista y opiniones personales de los ponentes/autores y no representan ni reflejan necesariamente los puntos de vista ni opiniones de Leica Biosystems, sus empleados o sus agentes. Cualquier enlace incluido en el contenido que proporcione acceso a recursos o contenido de terceros se proporciona únicamente por comodidad.
Para el uso de cualquier producto, debe consultarse la documentación correspondiente del producto, incluidas las guías de información, los prospectos y los manuales de funcionamiento.
Copyright © 2026 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.