
Transforming Pathology Services at Pathology & Cytology Laboratories, Kentucky
Driving Innovation and Unlocking Exceptional Productivity with Leica Biosystems BOND-PRIME IHC/ISH stainers and Aperio GT 450 DX scanners.
Within a month of deploying innovative technology from Leica Biosystems and embracing Universal Access workflows, Pathology & Cytology Laboratories were able to move away from a traditional, bottleneck-prone, batch-processing methodology to a fully digital, continuously flowing patient diagnosis and reporting paradigm.
Read how Kentucky Pathology & Cytology Laboratories (P&C) unlocked exceptional productivity gains, increased their diagnostic capacity, and reduced diagnostic turnaround times for patients by embracing Universal Access workflows and innovations.
The Solution: Digital Transformation and Workflow Innovation
The installation of Leica Biosystems BOND-PRIME IHC/ISH stainers and Aperio GT 450 DX slide scanners, together with a cloud-based LIS, has enabled ongoing, case-by-case immunohistochemistry (IHC) slide processing and instant digital reporting.
Initially, five Aperio GT 450 DX scanners were set up, showing they could reliably and quickly scan the daily workload. With an average scanning time of two minutes per slide and another two minutes for cloud LIS upload, slides are available for pathologist review within minutes of processing.
By introducing BOND-PRIME stainers and leveraging a continuous case workflow throughout the day, IHC slides can be completed in smaller batches on the same day as ordered. Thanks to the capacity of the Aperio GT 450 DX scanners, these slides are scanned right away and are ready for digital review as soon as they’re finished.
The Results: The Lab’s Just-in-Time Processing Model is Now a Benchmark for Productivity
Workflow changes from large batch to continuous production have lowered the need for overnight and early-morning shifts. The new system also eliminated the time-consuming manual slide sorting and distribution that was formerly necessary to deliver slides to the ordering pathologists.
Within four weeks of implementation, the lab delivered a staggering 88% improvement in next-day diagnostic turnaround times (TAT) – with 57% of all cases completing in just one day. The two-day TAT also improved, with 74% of cases completed in just two days. The improvement was most noticeable with remotely staffed pathologists who formerly had to wait an extra day for add-on recuts, special stains, and IHCs.
Elimination of outward-bound slide couriers allowed route reassignments, enabling expansion of service coverage to new opportunities or improving services within the current client base.

These improvements provide the organization with the assurance to confidently pursue expansion, knowing that an increased volume can be effectively accommodated.
The Challenge:
The laboratory operated on a 24/6 schedule, maintaining continuous staffing throughout the week except for just a few hours on Sunday. Batch IHC staining was performed mostly overnight, ensuring that slides were ready for distribution each morning.
At the start of each day, a dedicated team of five staff members sorted the slides that had been processed overnight, both hematoxylin and eosin (H&E) and IHC slides. After sorting, the staff collated the cases and organized the numerous slide trays for distribution. These trays were then delivered either directly to the offices of onsite Pathologists or sent via couriers to remote Pathologists working in medical centers across the state. This led to delays: onsite Pathologists received slides later because of courier priorities; offsite Pathologists already waiting on courier deliveries for their cases, also had to wait until the next day for additional slide requests, causing diagnostic delays.
The early morning slide-sorting shift was demanding and uninspiring. Along with the overnight shift, it experienced high staff turnover and open positions were difficult to fill. Replacing retiring pathologists within the Lexington office, let alone remote rural hospitals P&C covered, was becoming harder to do. Attracting and retaining specialist pathologists was difficult.
P&C aimed to revolutionize how pathologists and laboratory staff interact with diagnostic resources and information, seeking improvements in patient care, service quality, and cost efficiency. Their goal was not just to adapt, but to take the lead through innovative approaches.
Future-Proof Operations:
The implementation of continuous workflows has significantly enhanced productivity, enabling the laboratory to function with up to 48% surplus capacity without adding staff.
The adoption of new technologies, including Aperio GT 450 DX slide scanners and BOND-PRIME IHC stainers has enabled more efficient slide processing and diagnosis, allowing an improvement of 88% in next-day case completion and a further improvement of cases completing within two days.
Pathologists routinely complete reporting of cases by 2 PM. Remote pathologists were able to report a greater number of cases per day due to the digital availability of all work on demand. Pathologists attending off-site procedures can review cases during wait times.
Offering flexible working conditions has created an attractive environment for recruiting and retaining pathologists.

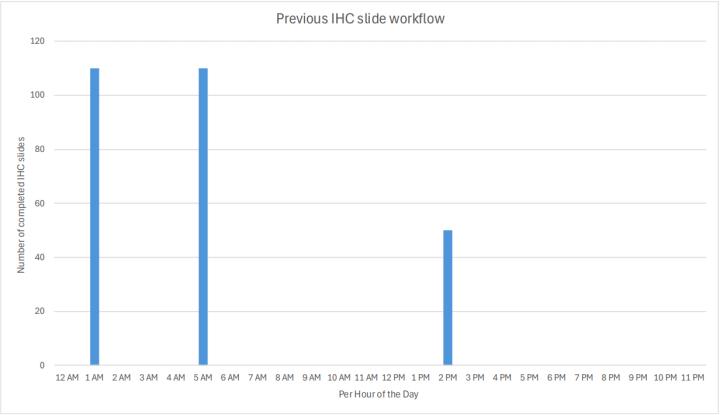
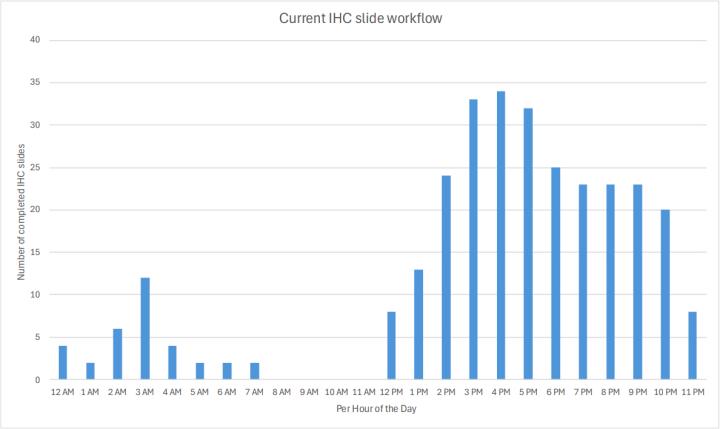
As shown in Graph 1, the lab previously prepped and saved slides from each day’s IHC orders, processed them overnight, and they were delivered to Pathologists the next day. The afternoon IHC run was for STAT orders only. This “big batch” concept overwhelmed the IHC stainers for short periods of time, but they then sat idle for the remainder of the day. To enable growth, if this workflow had been maintained, P&C would have needed more stainers. After moving to the two BOND-PRIME stainers and implementing continuous preparation and processing of IHC orders (Graph 2), P&C now has the capacity for more IHC slide volume throughout the day.
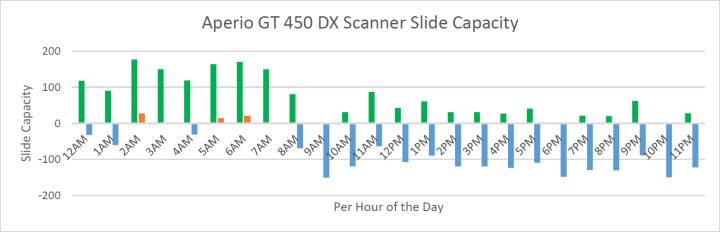
Graph 3 shows that the total volume of slides produced from midnight have completed scanning by 8 AM, with slides from 10 AM representing the add-on orders (deepers, recuts, and same-day IHC delivery). During the overnight hours, the digital scanners are mostly at full capacity (green), with available capacity (blue), and occasional periods of overcapacity when slides waiting to be loaded on a scanner (orange). Across the entire day, the green bars (representing digitally scanned slides) total 1700 slides, and the excess capacity (represented by the blue bars) totals 1800 slides. This means that the excess capacity of the Aperio GT 450 DX digital scanners is now at approximately 50% with a safety margin of 10-20%.
Collectively, these graphs indicate that the current scope of Leica Biosystems instrumentation can be scaled up by nearly 100% without necessitating changes to staffing levels.
A Strong Partnership Driving Laboratory Excellence:
P&C Laboratories and Leica Biosystems have built a longstanding, productive partnership that has contributed to enhancing the laboratory’s histology workflow. This collaboration is evident through the integration of multiple premium Leica Biosystems products (including HistoCore PELORIS 3 Tissue Processors and HistoCore SPECTRA Workstations) across key laboratory processes. Nearly 90% of IHC slides are processed using the BOND automated IHC/ISH stainers from Leica Biosystems. The Aperio GT 450 DX scanners complete a Leica Biosystems portfolio which spans the entire anatomic pathology workflow. These technologies, combined with Leica Biosystems’ expertise, support P&C’s commitment to delivering high-quality, accurate diagnostics and reinforce their dedication to continuous improvement through innovative solutions.

Who Are Pathology & Cytology Laboratories:
Pathology & Cytology Laboratories (P&C), located in Lexington, Kentucky, has provided diagnostic services since 1967. With a reach spanning 40 hospitals, 6 surgery centers, and over 1700 in-state medical offices and 1500 national Oral Surgeon offices, P&C’s mission is to deliver accurate, timely diagnostics—with a healthcare focus that is inclusive of rural and underserved communities.
The lab processes an average of 600 cases daily, generating 1700 slides (including approximately 250 IHC slides), and its workflow continues to grow. Its 18 pathologists, with 6 working remotely, now leverage digital slides for flexible reporting—even during off-site procedures. By enabling remote work, these digital capabilities have attracted top talent, including two of the nation's eight leading oral pathologists—and expanded specialty services.
P&C Labs run their own Histotech and Cytotech training programs, and the five former slide-sorting staff have transitioned into these programs, opening new career pathways.
Pathology & Cytology Laboratories’ journey demonstrates how workflow transformation and innovation can overcome rural healthcare barriers, empower staff, and deliver faster, higher-quality diagnostics. Their story is a testament to the power of partnership, technology, and a commitment to serving communities—urban and rural alike.
This case study was created in collaboration with the team at Pathology & Cytology Laboratories, Lexington, Kentucky. We’d like to express our particular thanks for their contributions and use of photography.
We congratulate Steve Olsen (pictured), outgoing COO, on his inspiring career in laboratory medicine and wish him all the best in retirement.


“When you initiate an innovation, it sparks innovation across the business” ― Steve Olsen
FOR IN VITRO DIAGNOSTIC USE
These results are applicable for the scenarios described here for this laboratory and cannot be used as a standalone claim. Individual results may vary.
These products may not be available in your country or region at this time. Please contact your Leica Biosystems sales representative or distributor for more information.
Leica Biosystems content is subject to the Leica Biosystems website terms of use, available at: Legal Notice. The content, including webinars, training presentations and related materials is intended to provide general information regarding particular subjects of interest to health care professionals and is not intended to be, and should not be construed as, medical, regulatory or legal advice. The views and opinions expressed in any third-party content reflect the personal views and opinions of the speaker(s)/author(s) and do not necessarily represent or reflect the views or opinions of Leica Biosystems, its employees or agents. Any links contained in the content which provides access to third party resources or content is provided for convenience only.
For the use of any product, the applicable product documentation, including information guides, inserts and operation manuals should be consulted.
Copyright © 2026 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.