
Techniques to Minimize Articular Cartilage Folding
In my department, we are not only interested in the histology of bone, but also in studying the articular cartilage of joints. When evaluating articular cartilage, look for any surface damage that has occurred from normal wear and tear or surgical models. After a joint has been decalcified, the loss of proteoglycans in the articular cartilage causes it to fold and ripple during the staining process. When this occurs, it makes it impossible to perform histomorphometry or measurements of the cartilage. In this article, I will explain some helpful tips to reduce or eliminate folding of the articular cartilage.
Grossing/Fixation
Every step in the histology process is critical, from dissection up to and including coverslipping, especially for articular cartilage samples. It is very important that your samples are dissected properly. For example, a mouse hind limb should be dissected well, leaving as little remaining soft and connective tissue as possible. When grossing around the joint, tissue is left intact to protect the joint (Fig. 1). If samples are incorrectly grossed or not kept hydrated, they will neither decalcify nor process well, leading to bad results. Bone samples should be fixed for a minimum of 72 hours in 10% Neutral Buffered Formalin. Samples are then rinsed well to remove the salts from fixation and then placed into a decalcifying agent, such as EDTA, for 7 to 14 days, depending on the sample and its size. The decalcifying agent should be fresh and changed regularly. It is crucial to use a decalcification end point test, such as x-ray, chemical testing using ammonium oxalate1 or weight test 2. It is imperative to remove all the calcium from the bone and equally important to not leave your samples in decalcification solution too long. Leaving samples in decalcifying agents too long causes excessive loss of the proteoglycans, increasing the odds of your sections folding.

Processing
Bone processing programs require longer processing cycles, generally 1 hour per station in automated processors. Larger samples, such as rabbit and dog need longer programs. The Animal Processing Manual from the National Society for Histotechnology, Veterinary, Industry and Research Committee is a valuable reference for processing all types of tissue 3. We use paraffin with higher concentrations of plastic polymers (Paraplast Plus) when processing, as it infiltrates bone tissue better. Tissue must be completely fixed before processing. A desired sample thickness is 3-4 mm, or about the thickness of a penny. If a sample is too large and "stuffed" into a cassette, the reagents will not be able to dehydrate, clear or infiltrate the tissue.
Embedding
Paraffin embedding is a critical step in the histological process when working with bone and joint samples, as it is with any sample. The orientation of your sample is very important. If bone is embedded at an angle, it may cause the bone to break and “chunk out” of the block. Harder paraffin (Paraplast) makes sectioning bone easier; it allows more support and stability. Embed tissue as flat as possible. If there is a cut side, embed that cut side down in the mold. Embed mouse leg, femur, and tibia in a “smile” orientation with the tibia/fibula connection up, as we are focusing on the skeletal muscle (Fig. 2).
Microtomy
Make sure you have a heavy-duty microtome when cutting any type of bone sample. When trimming or facing into your block, be very careful! Trim slowly and change the blade when necessary. If you trim in too fast, you can cause the bone or joint to chunk out. Put your block on an ice tray with a thin layer of water and soak your block before cutting. The colder the block, the easier bone is to section. When a block is cold, the paraffin is stronger, offering more support. When sectioning, if calcium remains in your sample it may cause the sample to chunk out or the section to lift from the slide, increasing the chances of articular cartilage folding. Bone and joints are difficult to get to adhere to slides. Slide selection should be considered carefully; a slide that is more charged will decrease the odds of folding and rippling. Because bone can be tricky to embed, you must angle your chuck/block holder in order to get to the proper level or plane. Be patient!
Staining
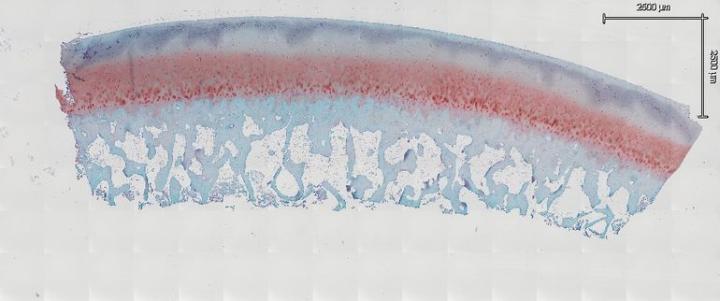
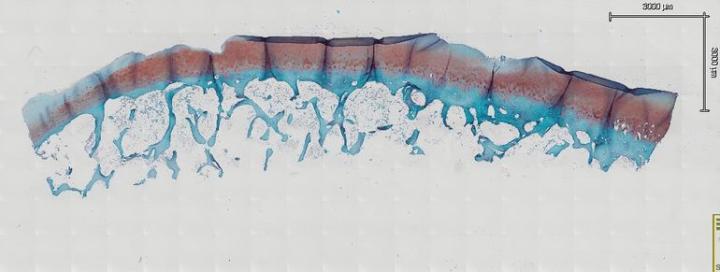
When preparing your sections for routine or special staining, you must take special care to prevent articular cartilage folding. Generally, we bake our slides overnight at 60° C to remove paraffin as well as adhere tissue to the slides. However, it has been found this is not the case with samples that have articular cartilage. In fact, after baking overnight, the probability of folding increases greatly. Allow sections to completely air dry overnight at room temperature. After drying, place the slides horizontally on a slide dryer at 37° C for a minimum of 12 hours. Many times, slides are left on the slide dryer over the weekend. All of our staining is done manually, which allows us to handle our samples in the gentle manner they require. If you move too fast or jar the slides, it may cause bone and joints to fold and lift from the slide. We then run the slides through a series of xylene, 100% EtOH, 95% EtOH, and 70% EtOH each for 5 minutes to dehydrate our samples. After dehydrating the samples, leave them at room temperature for 30 minutes to completely dry before staining (Fig. 3a and Fig. 3b). In our experience, this is the key step to reduce articular cartilage folding. After your section is completely dry, proceed to your stain protocol as normal. The other key factor to prevent articular cartilage folding is to air dry your sections again after a series of 70% EtOH, 95% EtOH, and 100% EtOH for the rehydration step. After the slides are completely dry, dip them in xylene and coverslip.
The same steps used in the dehydration and rehydration processes can be used while performing immunohistochemistry to avoid articular cartilage folding.
Immunohistochemistry
Immunohistochemistry protocols that require heat-induced antigen retrieval can be damaging to both the cartilage and bone. High temperatures can cause the bone and joint to lift off the slide completely or to ripple causing artifacts. If an immunohistochemistry protocol calls for using heat retrieval, lower the temperature and extend the incubation time.
When using histomorphometrically to measure articular cartilage following injury or surface damage, the articular cartilage cannot be folded. Folding of the cartilage will give measurements that are neither accurate nor true representations of your sample.
Careful and gentle handling of articular cartilage samples and sections is essential. Following proper histological steps from dissecting your tissue to cover slipping your slide are critical to obtaining quality articular cartilage sections.
This presentation was prepared by Sarah Mack, Manager Histology at the University of Rochester, NY.
About the presenter

Sarah A. Mack has over a decade of experience as a histotechnician, with a main focus on preparation, processing and analysis of both calcified and soft tissues of the musculoskeletal system. In addition to providing consulting, advice and assistance to clients of the HBMI Core, Sarah serves as its Manager, supervising all histology-based projects within the Center of Musculoskeletal Research. Sarah supervises the generation of musculoskeletal tissue biorepositories, and oversees the creation and maintenance of reagent stocks and standard protocols. She provides hands-on assistance in the preparation of histological tissues, including decalcification, automated paraffin processing, orientation before embedding and sectioning of paraffin and frozen tissue.
Sarah currently serves as Chair of the Hard Tissue Committee for the National Society of Histotechnology and as President of the New York State Histotechnological Society. She has been an active member in both societies for more than 15 years, providing information and knowledge to histologists that work in both scientific and clinical lab settings.
References
- Skinner RA, Hickmon SG, Lumpkin CK, Aronson J, Nicholas RW. Decalcified Bone: Twenty Years of Successful Specimen Management. The Journal of Histotechnology 1997;20;267-277.
- Callis G, Sterchi D. Decalcification of Bone: Literature Review and Practical Study of Various Decalcifying Agents, Methods, and Their Effects on Bone Histology. The Journal of Histotechnology 1998;21;49-58.
- Animal Processing Manual. First ed., National Society Of Histotechnology, 2002.
Related Content
Leica Biosystems Knowledge Pathway content is subject to the Leica Biosystems website terms of use, available at: Legal Notice. The content, including webinars, training presentations and related materials is intended to provide general information regarding particular subjects of interest to health care professionals and is not intended to be, and should not be construed as, medical, regulatory or legal advice. The views and opinions expressed in any third-party content reflect the personal views and opinions of the speaker(s)/author(s) and do not necessarily represent or reflect the views or opinions of Leica Biosystems, its employees or agents. Any links contained in the content which provides access to third party resources or content is provided for convenience only.
For the use of any product, the applicable product documentation, including information guides, inserts and operation manuals should be consulted.
Copyright © 2026 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.



