What Is Translational Research?


This article is the second of a 4-part series including the following topics:
“What is Life Science”, “Translational Research” (current feature), “Spatial Research,” and “Biomarkers in Translational/Spatial Research.”
Translational research, also cited as translational science or translational medicine, is a newer field aimed at producing result-oriented data to be utilized for the direct benefit of society. The last 4 decades have seen substantial activity in translational research (TR). TR is still in its infancy, as the term started to gain traction only in the early 90s1 however, as emerging technologies are refined to identify precise targets in early stages of product development, the importance of TR will continue to rise in the scientific community and the global population. This article discusses the TR process, its subcategories, its overall impact, and future challenges.
1. What is Translational Research?
Translational research is a field that investigates potential application of a scientific idea to directly benefit human beings and improve lives. It acts as a bridge between basic and clinical research by analyzing the value of newly discovered scientific ideas for use in the clinical setting. This field requires interdisciplinary collaboration, with investigators from bioinformatics, biostatistics, molecular biology, and oncology.1 TR is a relatively new and developing field which, since its inception, has continuously gained momentum in the scientific community. TR is divided into phases capturing specific product developmental stages, labeled T0, T1, T2, T3, and T4. As reflected in these stages, TR entails rigorous planning to bring a product from essential idea generation to practical application (Figure 1).
In addition to the core interdisciplinary team of collaborators, evidence generation across the phases of TR may require input from other experts, including spatial biologists, computational pathologists, histotechnologists, and geneticists, depending on research needs. The data from each expert is analyzed, and at every stage, the practical value of the evidence is weighed against the risk-benefit ratio. A single TR program, from T0 to T4, may require the contribution of 20 different scientific disciplines in the discovery-patient trial-clinical practice pipeline.2A TR study could take around 12-15 years and billions of dollars, and chances are that the tested product may not be proven safe and effective, as data shows less than 1% of the TR is successful.2 To improve translational research's predictability, leading global organizations such as the National Institutes of Health have set up dedicated translational science institutes. National Center for Advancing Translational Sciences is an NIH wing founded specifically to increase the proportion of successful TR study outcomes. Similarly, European Advanced Translational Research Infrastructure (EATRIS) is also focused on translational research advancement.3
Goals
Out of the 8000 diseases known to affect human beings, only 600 have approved treatment.2 Hence, in addition to identifying a disease's pathophysiological properties using basic science, application of that knowledge toward finding a treatment that could be used in clinical medicine is essential. The primary goal of TR is to have a strategic workflow from product discovery to a ready-to-use solution that may aid diagnosis and/or treatment for the broader human population. TR uses a planned approach to work on a product that would specifically benefit the human population.
The risk of losing billions of dollars in TR is high, since less than 0.1% of planned drug products pass the rigors of clinical testing and are deemed fit to be used by the community.4 Therefore, effective TR should use an open innovation and collaborative approach leveraging the disciplines of spatial biology, genomics, computational biology, and proteomics to identify roadblocks so that corrective actions can be taken to rectify the research course in a timely manner. In this way, a newer goal of TR is to save time and resources while continuously developing scientific innovation for human health.
2. Relationships Among Translational, Basic, and Applied Research
Translational, basic, and applied research are interconnected and essential for the successful execution of the therapeutic development process (Table 1). Basic research is where an innovative idea originates, whereas applied research takes that idea forward and explores the possibility of incorporating it into clinical practice. Translational research includes both basic and applied research elements to understand the practical application of the scientific idea. Basic research undertakes the responsibility to answer fundamental questions and gather novel information about a disease which may not be directed toward any immediate practical use, while Applied research investigates a problem that attempts to find a real-world practical solution to a problem which may benefit the society in near future. TR takes concrete steps to move scientific knowledge from discovery to patient care for improved health outcomes.
Table 1. Compare and contrast between Translational, basic, and applied research
| Translational Research | Basic Research | Applied Research |
|---|---|---|
| Act as a bridge between basic and applied research to find innovative interventional treatment for societal benefit | Generates scientific discovery ideas that may include preclinical models or in vitro cell- based studies | Focuses on finding practical real-world solution related to an indication rather than studying a particular disease from start to finish |
| Clear “bench to bedside” strategy- focuses on converting scientific knowledge to improve human lives | Foundation for applied and translational research | Research conclusions primarily addresses solutions to immediate practical problems |
3. Translational Research and Clinical Research
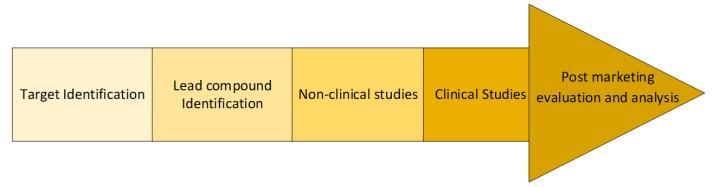
Translational research aims to incorporate scientific innovation studies at the laboratory or clinical level by comprehensively testing an idea to become suitable for diagnostic, interventional, or any other treatment to enhance clinical medicine.5 Translational research is defined as converting a basic scientific observation from the laboratory, clinical medicine, or the broader population into therapeutic interventions or diagnoses to advance human health.6 Clinical research involves studying a scientific concept/idea specifically in humans. Clinical research via observational or behavioral studies tests the safety and effectiveness of an investigational product in a human population. To confirm preclinical safety and efficacy results, clinical trials are performed on humans. Clinical trials are conducted in 4 phases; Phase I, Phase II, Phase III, and Phase IV. Phase I is conducted in around 50 healthy volunteers to test the safety. Phase II (around 100 patients) and III (around hundreds of patients) are large scale trials to assess the efficacy. After Phase III trial is successfully completed, the product is approved by the regulatory agency. Phase IV trials (around thousands of patients) continue to monitor the long-term side effects or additional benefits of the product to the patients.5,7
Clinical research is an integral part of translational research in which the safety and efficacy of a diagnostic or a therapeutic product are analyzed. In the transition from the non-clinical to the clinical phase, 50% of investigational products (IP) fail without going to the later stages of clinical trials. Since the failure rate of IPs at the initial stages is significantly high, this stage is also termed the “Valley of Death”.4 Public and private funding are essential to cross this divide and continue clinical projects. With evolving technologies, researchers have developed techniques such as “clinical trial in a dish” (CTiD) to mitigate possible risks before testing IPs in critical clinical trials. In CTiD, cells from human tissues are used to test/screen new chemical entities in human tissues. This acts as a bridge between the preclinical and clinical assessment of investigational products. Table 2 summarizes the contrasts between the 2 branches.
Table 2. Differences between translational and Clinical research
| Translational Research | Clinical Research |
|---|---|
| Focuses on problems that will directly benefit society | The outcome may or may not benefit society |
| Utilizes several cross-functional collaborators, including pharmacologists and epidemiologists | May not always involve extensive collaboration |
| Utilizes basic (preclinical animal models, in vitro laboratory research) and clinical research to meet goals | Does not include basic research |
| Incorporates elements from basic science to preclinical and clinical trials to empirical studies to meet its objective of improving health outcome | In-human studies in controlled clinical settings to test the investigational product’s safety and efficacy |
4. Process
Translational research is divided into T0, T1, T2, T3, and T4 stages,4,6 in an iterative process in which researchers are open to any opportunity to fine-tune a previous phase if the current phase fails or does not perform optimally.
Each phase of TR progresses a scientific idea from discovery to realizing its potential for actual use in improving human health, by comprehensively testing and evaluating the safety and efficacy of a diagnostic, drug, or medical device. As reflected in the multiple phases of the translational research spectrum, product testing, optimization, and testing in different clinical settings is a lengthy process that requires foresight and continuous improvement. A typical translational research model is described in Figure 2.
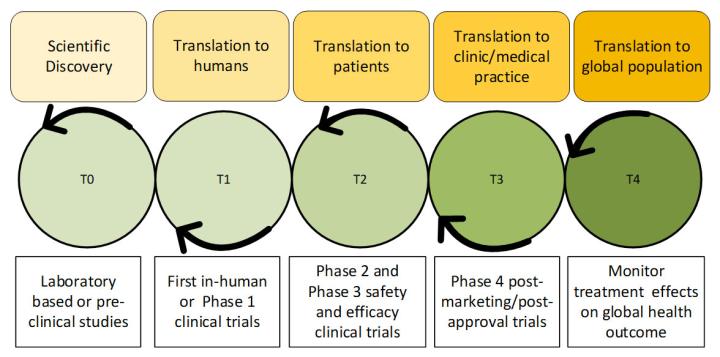
4.1 T0 phase
The T0 phase is defined as the conceptualization phase including scientific discovery, which is the foundation of the T1 to T4 stage.8 In this phase, a problem is identified via preclinical study or in vitro laboratory experimentation. For example, “Antibody X targets gene Y to inhibit angiogenesis” is an observation that may emanate from T0 research. This phase may also require elaborate non-clinical testing to reach its conclusion, including toxicity studies, carcinogenicity testing, and other such analyse that will ensure product safety for human testing.
4.2 T1 phase
T1 determines the potential application of the T0 observations for clinical use, or the pharmacological potential of an investigational product for humans. The T1 phase generally consists of proof of concept (PoC) studies in clinical trials involving small cohorts of approximately 30-50 volunteer subjects to test toxicity and other parameters. This may include analyzing an investigational product's pharmacokinetics (what the drug does to the body) and pharmacodynamics (what the body does to the drug).9
4.3 T2 phase
The T2 stage utilizes a more extensive setting to measure the efficacy (ability of a product to produce a desired effect) of an investigational product in a more comprehensive cohort of human subjects. This includes phase 2 and phase 3 clinical trials, which are conducted in 500-1000 patients to generate evidence of product approval. This type of testing in a randomized, double-blind trial can inform the investigators of the product's efficacy.
The T2 phase evaluates the potential of an investigational product in a controlled setting and investigates/confirms the therapeutic or diagnostic potential. A specific regulatory agency, such as the FDA, approves the tested therapy if the data from this phase shows promise. After the approval, new guidelines are drafted to educate the medical community on using the new therapeutic product.
4.4 T3 phase
The T3 phase includes analyses that evaluate whether a therapeutic product is suitable for medical practice. These studies include post-market phase 4 clinical trials conducted after gathering additional information, which may encompass risk mitigation, side effects, and new indications. The therapy under study could show side effects in this phase which did not surface during earlier testing with a limited sample size. If the side effect is significant, the therapy is withdrawn immediately. Researchers may also compare how a new therapy fares in terms of cost and effectiveness against comparator products.
4.5 T4 phase
The T4 phase assesses the effect of the intervention on the global human population.10 In this phase, the value of the drug/device to the broader population is studied via epidemiological analyses. Researchers involved in T4 studies will continue to monitor data from global post-market phase 4 trials and delve deeper into disease outcomes, assessing treatment benefit from real-world data and performing multi-center studies to evaluate global side effects. Additionally, public policy may be revised to ensure that the product is accessible to a wider socioeconomic range of patients.
5. Challenges
TR has substantial potential; however, most clinicians find doing TR or clinical research more challenging than basic research. TR is more expensive than basic research as it involves testing ideas in complex organisms; including animal models, ultimately transitioning to extensive human trials.4 Additionally, there is a dearth of trained clinical and translational research scientists.11Recently, there have been several programs, such as the Clinical and Translational Science Award (CTSA), to support translational scientists.11 Funding programs like the CTSA should continue to be developed to encourage TR and widen access to researchers across the world.
However, primary funders such as the National Institutes of Health must be careful to strike a balance when funding basic or applied research. Applied research or translational research may be an attractive option for the funders, since the clinical benefit is clearer. Still, basic science answers the foundational questions that clinical and translational research leverage for human studies. The past decade has seen a significant increase in funding for translation and clinical research, with a narrowing focus on basic science.12 Therefore, care must be taken not to neglect fundamental research. To counter an imbalance of funding and a future lack of fundamental research programs, academic and industrial translational scientists should work together to produce the most benefit to the scientific community and the public.
6. Conclusion
Translational research is an important field that will continue to grow if appropriate academic and industrial community support is provided. Although the attrition rates are significantly high among researchers in TR, the value of delivering novel treatment is immense. Since this branch is comparatively new, the scientific community and funding organizations must open avenues to support and encourage investigators interested in making TR their career path. Since a successful TR demands extensive collaboration, researchers should have an open innovation translational approach emphasizing transparent communication of ideas to optimize the pipeline of scientific ideas, from the “seed” of hypothesis to the “fruit” of a viable actionable diagnostic product.
About the presenters

Shubham Dayal is a Senior Medical Writer at Leica Biosystems and has over 10 years of experience in regulatory/preclinical/clinical writing for studies that are at different stages of the product lifecycle. Shubham has a PhD in Cell and Molecular Biology from the University of Toledo and a Master's in Regulatory Affairs from Northeastern University and has co-authored multiple peer-reviewed articles and poster presentations. He is an active member of the Regulatory Affairs Professional Society and American Medical Writers Association and holds certifications related to scientific writing. In his current role, Shubham's goal is to create awareness for our customers in ways that can advance scientific communication and ultimately improve patient outcomes.

Jack obtained his doctorate in molecular and cellular pathology and performed post-doctoral studies on cancer epigenetics and cardiovascular post-translational modifications. He has worked in externally facing roles at Leica Biosystems for 7 years, and currently works in partnership with leading pathologists and researchers to advance scientific study at the cutting edge of anatomic pathology research.
References
- Strand, D.L. Everyday characterizations of translational research: researchers’ own use of terminology and models in medical research and practice. Palgrave Commun 6, 110 (2020). https://doi.org/10.1057/s41599-020-0489-1.
- Austin CP. Opportunities and challenges in translational science. Clin Transl Sci. 2021;14(5):1629-1647. doi:10.1111/cts.13055 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8504824/#:~:text=The%20translational%20process%20is%20seen,exactly%20what%20would%20be%20expected.
- van Dongen GA, Ussi AE, de Man FH, Migliaccio G. EATRIS, a European initiative to boost translational biomedical research. Am J Nucl Med Mol Imaging. 2013;3(2):166-174. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3601476/
- Seyhan, A.A. Lost in translation: the valley of death across preclinical and clinical divide – identification of problems and overcoming obstacles. transl med commun 4, 18 (2019). https://doi.org/10.1186/s41231-019-0050-7.
- SC CTSI. What is Clinical & Translational Science. https://sc-ctsi.org/about/what-is-clinical-translational-science
- ICTR. What are the T0 and T4 Research Classifications? https://ictr.wisc.edu/what-are-the-t0-to-t4-research-classifications/
- MICHR. WHAT IS CLINICAL & TRANSLATIONAL RESEARCH? https://michr.umich.edu/what-is-clinical-translational-research
- UAMS; Translational Research Institute. What is Translational Research? https://tri.uams.edu/about-tri/what-is-translational-research/
- American Cancer Society. Types and Phases of Clinical Trials. https://www.cancer.org/cancer/managing-cancer/making-treatment-decisions/clinical-trials/what-you-need-to-know/phases-of-clinical-trials.html
- Lopez-Class M, Peprah E, Zhang X, Kaufmann PG, Engelgau MM. A Strategic Framework for Utilizing Late-Stage (T4) Translation Research to Address Health Inequities. Ethn Dis. 2016;26(3):387-394. Published 2016 Jul 21. doi:10.18865/ed.26.3.387. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4948806/
- Sweeney C, Schwartz LS, Toto R, Merchant C, Fair AS, Gabrilove JL; CTSA Mentored-to-Independent Investigator Transition Working Group. Transition to Independence: Characteristics and Outcomes of Mentored Career Development (KL2) Scholars at Clinical and Translational Science Award Institutions. Acad Med. 2017 Apr;92(4):556-562. doi: 10.1097/ACM.0000000000001473. PMID: 28351069; PMCID: PMC5373479. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5373479/
- Fang FC, Casadevall A. Lost in translation--basic science in the era of translational research. Infect Immun. 2010;78(2):563-566. doi:10.1128/IAI.01318-09. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2812192/
Related Content
Leica Biosystems content is subject to the Leica Biosystems website terms of use, available at: Legal Notice. The content, including webinars, training presentations and related materials is intended to provide general information regarding particular subjects of interest to health care professionals and is not intended to be, and should not be construed as, medical, regulatory or legal advice. The views and opinions expressed in any third-party content reflect the personal views and opinions of the speaker(s)/author(s) and do not necessarily represent or reflect the views or opinions of Leica Biosystems, its employees or agents. Any links contained in the content which provides access to third party resources or content is provided for convenience only.
For the use of any product, the applicable product documentation, including information guides, inserts and operation manuals should be consulted.
Copyright © 2026 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.