
Bond Oracle HER2 IHC System
Marketing Approval Number for In Vitro Diagnostic 22400AMX00674000
Brand Name Bond Polymer System HER2 Test
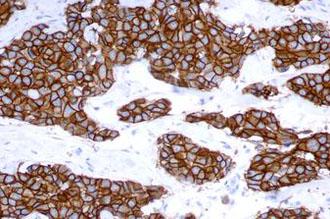
The Bond Oracle HER2 IHC System is a fully-automated assay to accurately determine HER2 oncoprotein status in breast and gastric cancer tissue as an aid in the assessment of patients for whom Herceptin® (trastuzumab) treatment is being considered.
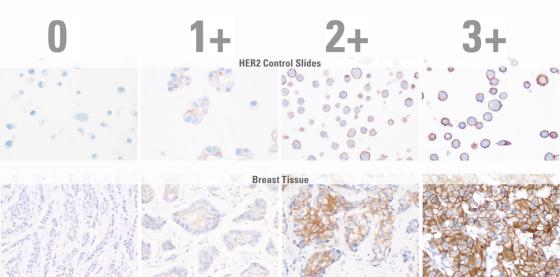
- Accurate - optimized to precisely separate 1+ and 2+ cases gives you excellent FISH concordance, reducing avoidable FISH requests.
- Drive Consistency - standardized protocols for uniform staining, decreased process variation, and consistent results.
- Maximize Efficiency - full automation frees up your skilled staff for other high-value tasks.
- A Complete Solution - integrated ready-to-use reagents, control slides, and advanced automation ensure quality and consistency.
For In Vitro Diagnostics Use
Product Features
Diagnostic Confidence
With treatment decisions dependent on a stained slide, you need confidence that your HER2 staining for breast and gastric cancer is consistent and accurate. The Bond Oracle HER2 IHC System gives you the assurance that comes with demonstrated HER2 FISH concordance and complete assay validation.
With the Oracle system, you get the accurate results needed for effective patient management.
Laboratory Efficiency
‘Go Lean’ without increasing resources. The Bond Oracle HER2 IHC System and the fully automated BOND-MAX and BOND-III staining platforms improve the efficiency of your laboratory with increased productivity and walk-away convenience.
Brochure
 Oracle HER2 IHC System Brochure Breast and Gastric
Size: 1.28 MB,
Type: pdf
Oracle HER2 IHC System Brochure Breast and Gastric
Size: 1.28 MB,
Type: pdf
 Oracle HER2 Interpretation Guide (Breast)
Size: 1.34 MB,
Type: pdf
Oracle HER2 Interpretation Guide (Breast)
Size: 1.34 MB,
Type: pdf
 Oracle HER2 Interpretation Guide (Gastric)
Size: 2.07 MB,
Type: pdf
Oracle HER2 Interpretation Guide (Gastric)
Size: 2.07 MB,
Type: pdf
 Oracle HER2 Scoring Guide (Breast)
Size: 729.56 KB,
Type: pdf
Oracle HER2 Scoring Guide (Breast)
Size: 729.56 KB,
Type: pdf
 Oracle HER2 Scoring Guide (Gastric)
Size: 498.97 KB,
Type: pdf
Oracle HER2 Scoring Guide (Gastric)
Size: 498.97 KB,
Type: pdf
Material Safety Data Sheets
 TA9145 MSDS BG 08JAN2021
Size: 1.09 MB,
Type: pdf
TA9145 MSDS BG 08JAN2021
Size: 1.09 MB,
Type: pdf
 TA9145 MSDS DA 08JAN2021
Size: 849.26 KB,
Type: pdf
TA9145 MSDS DA 08JAN2021
Size: 849.26 KB,
Type: pdf
 TA9145 MSDS DE 08JAN2021
Size: 947.27 KB,
Type: pdf
TA9145 MSDS DE 08JAN2021
Size: 947.27 KB,
Type: pdf
 TA9145 MSDS EL 08JAN2021
Size: 1.01 MB,
Type: pdf
TA9145 MSDS EL 08JAN2021
Size: 1.01 MB,
Type: pdf
 TA9145 MSDS EN 08JAN2021
Size: 926.33 KB,
Type: pdf
TA9145 MSDS EN 08JAN2021
Size: 926.33 KB,
Type: pdf
 TA9145 MSDS ES 08JAN2021
Size: 813.18 KB,
Type: pdf
TA9145 MSDS ES 08JAN2021
Size: 813.18 KB,
Type: pdf
 TA9145 MSDS FR 08JAN2021
Size: 942.3 KB,
Type: pdf
TA9145 MSDS FR 08JAN2021
Size: 942.3 KB,
Type: pdf
 TA9145 MSDS HU 08JAN2021
Size: 1.01 MB,
Type: pdf
TA9145 MSDS HU 08JAN2021
Size: 1.01 MB,
Type: pdf
 TA9145 MSDS IT 08JAN2021
Size: 950.16 KB,
Type: pdf
TA9145 MSDS IT 08JAN2021
Size: 950.16 KB,
Type: pdf
 TA9145 MSDS NL 08JAN2021
Size: 863.88 KB,
Type: pdf
TA9145 MSDS NL 08JAN2021
Size: 863.88 KB,
Type: pdf
 TA9145 MSDS NO 08JAN2021
Size: 842.56 KB,
Type: pdf
TA9145 MSDS NO 08JAN2021
Size: 842.56 KB,
Type: pdf
 TA9145 MSDS PL 08JAN2021
Size: 1.1 MB,
Type: pdf
TA9145 MSDS PL 08JAN2021
Size: 1.1 MB,
Type: pdf
 TA9145 MSDS PT 08JAN2021
Size: 859.31 KB,
Type: pdf
TA9145 MSDS PT 08JAN2021
Size: 859.31 KB,
Type: pdf
 TA9145 MSDS RO 08JAN2021
Size: 1021.53 KB,
Type: pdf
TA9145 MSDS RO 08JAN2021
Size: 1021.53 KB,
Type: pdf
 TA9145 MSDS RU 08JAN2021
Size: 1022.6 KB,
Type: pdf
TA9145 MSDS RU 08JAN2021
Size: 1022.6 KB,
Type: pdf
 TA9145 MSDS SL 08JAN2021
Size: 1.05 MB,
Type: pdf
TA9145 MSDS SL 08JAN2021
Size: 1.05 MB,
Type: pdf
 TA9145 MSDS SV 08JAN2021
Size: 839.11 KB,
Type: pdf
TA9145 MSDS SV 08JAN2021
Size: 839.11 KB,
Type: pdf
 TA9145 MSDS TR 08JAN2021
Size: 1 MB,
Type: pdf
TA9145 MSDS TR 08JAN2021
Size: 1 MB,
Type: pdf
 TA9145 SDS JP
Size: 2.17 MB,
Type: pdf
TA9145 SDS JP
Size: 2.17 MB,
Type: pdf