End to End Portfolio Solutions
Leica Biosystems strives to help our customers reach their objectives by connecting departments, solutions, and processes across the cancer diagnosis pathway.
Tissue Processing
Automated solutions to maximize laboratory productivity for efficient and flexible workflow.
Pre-Analytics & Specimen Preparation
Cryostats for high quality sectioning, user safety, and efficient workflow.
Versatile printing of tissue cassettes and slides allows quick and accurate case identification.
Specimen Processing
Automated tissue processors to maximize laboratory productivity for efficient and flexible workflow.
Modular tissue embedding system offers the flexibility to suit your laboratory.
Staining
Automated H&E staining ensures quality and consistency.
Fully automated IHC and ISH Stainers provide accurate and timely results.
Digital Pathology
High capacity, clinically validated digital solution drives quality and efficiency.
Case Studies
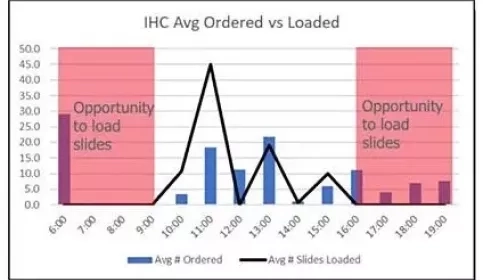
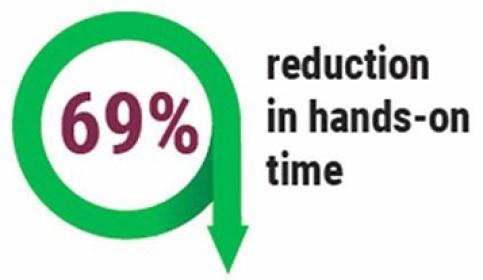
Optimization of Staff and Equipment to Boost Throughput
The Leica Biosystems Process and Solutions Optimization team has partnered with a major academic facility within an Integrated Network to examine how to optimize staffing and equipment in their immunohistochemistry lab.
Optimizing anatomical pathology through implementation of specimen tracking
The Leica Biosystems Process and Solutions Optimization team has partnered with a major academic medical center to examine ways to further optimize their anatomic pathology lab and equipment.
Case Base verses single piece IHC Workflows Evaluated in a large reference Laboratory
The Leica Biosystems Process and Solutions Optimization team has partnered with a high-volume reference laboratory to evaluate the potential impact of a case based IHC workflow versus an individual slide workflow.
Videos and Webinars
New study shows BOND-III up to 40% faster than Benchmark Ultra
In a recent study, the Leica Biosystems BOND-III was found to complete cases up to 100 minutes faster than the Roche Tissue Diagnostics BenchMark ULTRA...
Novocastra p16 antibody demonstrated an agreement of 98% with the Roche p16 antibody
The study compared the percentage agreement rates of Novocastra’s p16 (6H12) IVD solution with Roche CINtec® Histology p16 assay on 170 oropharyngeal cancer cases...
Single cells shine new light on lung biology and disease
Keith Wharton, MD PhD FCAP, Senior Medical Director for Leica Biosystems (Advanced Staining and Imaging) has over 30 years of experience in academic and bipharma settings...
Educational
An Introduction to Specimen Processing
Most fresh tissue is very delicate and easily distorted and damaged, and it is thus impossible to prepare thin sections from it unless it is chemically preserved or “fixed” and supported in some way whilst it is being cut. Broadly there are two strategies that can be employed to provide this support...
H&E Staining Overview: A Guide to Best Practices
For routine diagnosis, the use of Hematoxylin and Eosin (H&E) is by far preferred for viewing cellular and tissue structure detail by pathologists. The variation of stain intensity is often driven by the pathologist’s learning experience and personal preference...
An Introduction to Routine and Special Staining
Routine H&E staining and special stains play a critical role in tissue-based diagnosis or research. By colouring otherwise transparent tissue sections, these stains allow highly trained pathologists and researchers to view, under a microscope, tissue morphology (structure)...