
Biomarkers


FOR RESEARCH USE ONLY. Not For Use In Diagnostic Procedures.
One of the most exciting advancements in cancer diagnosis, prognosis, and treatment is detection of biomarkers.1 Detecting an appropriate biomarker may help decide the best therapy for certain cancers, and it can enable rapid decision-making to prolong the lives of cancer patients. Biomarkers are measured across many diseases, and they have a wide array of applications. By definition, a biomarker should be measurable, providing an objective assessment of the disease state. For example, blood sugar level measurements are biomarkers for hypertension or diabetes assessments. Similarly, a clinician can use biopsies to quantify biological markers such as EGFR that could guide treatment regimens for cancer.2
Biomarkers are vital for precision medicine and the study of the tumor microenvironment. Since the tumor microenvironment is heterogenous, biomarker expression may vary across samples and among patients with the same cancer type. Identification of which cells express certain biomarkers can provide information to enable a clinician to choose the most appropriate cancer treatment.3 For example, blood concentration of prostate specific antigen (PSA) is a reliable biomarker for prostate cancer and helps determine next steps for patients and clinicians.
According to the WHO, a biomarker is defined as “any substance, structure, or process that can be measured in the body or its products and influence or predict the incidence of outcome or disease.”4
A biomarker can be a gene, protein, or other chemical entity that can prognostically, diagnostically, or otherwise measure probability or predisposition to a disease state. A biomarker indicates a normal physiological process, pathogenic exposure, or external intervention. These can be extracted from bodily fluids, including blood, serum, or urine, and can be analyzed by measuring the DNA, RNA, or protein levels via Immunohistochemistry (protein), fluorescent in situ hybridization (DNA/RNA), or polymerase chain reaction (RNA). Since specific treatments target a particular subtype or disease state, biomarkers have become essential in informing clinicians whether the proposed treatment would benefit the testing cohort.5
A biomarker should be comprehensively validated through preliminary studies.
Types of Biomarkers
Susceptibility or risk biomarkers
This type of biomarker can give information about a healthy individual's susceptibility to developing a specific disease. The risk biomarker can be detected well before disease progression. Genetic variation in BRCA1/2 can be an early determinant of breast cancer risk, and mutations in the APOE gene can indicate an increased risk of developing Alzheimer's disease.6 These types of biomarkers provide valuable information on disease management or preventive surgery to avoid any prospective disease.7
Diagnostic Biomarkers
Diagnostic markers should be a signature indication of a disease. Hemoglobin HbA1c is regularly used as a signature diagnostic marker for diabetes mellitus.8 The molecular levels are increased with the corresponding increase in glycosylation. Diagnostic biomarkers are used to differentiate and confirm a disease state, which includes disease staging or identification of specific biological phenomena such as oncogenesis. In such cases, accuracy is paramount to reaching the correct diagnosis and preserving patient health. Therefore, each parameter of the diagnostic marker's detection assay must be validated via a statistically powered clinical study before testing it in routine clinical practice.1 In prostate cancer testing, where PSA is considered a diagnostic marker, a faulty result can not only jeopardize a patient's well-being but can also be psychologically and financially detrimental.
Prognostic and Predictive Biomarkers
Prognostic biomarkers
These biomarkers can predict the disease probability (progression or recurrence) after initial diagnosis. Prognostic biomarkers provide the information of the disease trajectory/outcome over a time-period (disease metastasis, tumor volume, etc.) irrespective of therapeutic intervention.9 For example, BRCA1/2 is a prognostic biomarker for breast cancer patients, and its elevated levels are associated with worse disease outcomes.10 Similarly, high expression levels of death receptors (DR5) are associated with worse outcomes in colorectal cancer cases,11 while IRF4 and EGR1 gene mutations are associated with favorable survival rate in multiple myeloma patients.12 The difference between susceptibility and prognostic biomarkers is that the susceptibility biomarker is not related to any change to the clinical treatment. In contrast, the prognostic marker can suggest possible clinical treatment based on disease progression and the corresponding biomarker’s expression status.9, 13
In many cases prognostic marker level is included as an exclusion/inclusion criterion for a study. These markers are beneficial in staging or stratifying patients for a clinical trial. Many molecules are related to disease progression. Therefore, a useful prognostic marker should be able to provide information to the clinician on the stage of disease progression so that an appropriate treatment regimen is provided. The prognostic biomarker may help suggest an appropriate treatment, but unlike predictive biomarkers cannot predict the treatment response rate.
Predictive biomarkers
These biomarkers help predict the type of clinical treatment most appropriate for a particular disease state. Biomarkers such as ER or HER2 can predict the way forward for disease management and treatment response assessment. ER and PR can be predictive as well as diagnostic.14 Evidence has suggested that these markers give information on who would not benefit from the treatment and not who would benefit from the treatment.
ER, for example, can be a predictive marker for those who won't benefit from breast cancer hormonal therapy. Still, it does not necessarily predict that everyone who will receive hormonal therapy will benefit from the treatment.5 EGFR is another predictive biomarker example for which Gefitinib inhibitor therapy is used to treat non-small cell lung cancer patients. A comparative analyses between the 2 types of biomarkers is provided in Table 1.
Table 1. Comparison between predictive and prognostic markers
| Predictive | Prognostic |
| Depends on the treatment response to the disease progression | Independent of the treatment response to the disease progression |
| Helps in the treatment decision-making process | Does not help in the treatment decision-making process |
| Status informs to the effectiveness of the treatment | Status independent of the treatment efficacy |
| Immune checkpoint inhibitors such as PD-L1, one of the predictive biomarker examples, can aid in screening patients that may benefit from a specific treatment | TP53 biomarker is one of the examples of prognostic biomarker that could inform clinicians of the patient's overall survival rate |
The difference, at times, between prognostic and diagnostic biomarkers is not stark; therefore, in some cases, one candidate molecule can act as prognostic and predictive (for example, ER, PR, VEGF, etc.).11
Monitoring Biomarkers
This type of biomarker helps monitor a change in a patient's health in response to an intervention or drug treatment. A molecule that can be measured periodically is a monitoring biomarker. A monitoring biomarker can also fall into one of the other biomarker categories. As long as the molecule can monitor a change in a disease state, it is called a monitoring biomarker. Molecules such as CA125 and PSA are diagnostic for ovarian and prostate cancer, respectively, since they can be measured and monitored periodically to analyze the disease state continuously.
Safety Biomarkers
Safety biomarkers are molecules used to assess the adverse effects of drug intervention or environmental exposure that may lead to systemic toxicity. For example, bile pigments bilirubin and biliverdin levels are observed to analyze hepatotoxicity.15
Response biomarkers
These biomarkers provide information on the treatment's adequacy (toxic or optimal new chemical entity) against a disease. In cancer, response biomarkers use a decrease in tumor size and progression-free survival as surrogate endpoints rather than relying on long-term overall survival rates as an endpoint, risking drug-related toxicities to the patient. Similar to other biomarker types, these biomarkers are expected to be detected at the lowest levels of drug response.5
Circulating tumor cells, circulating tumor markers, and circulating nucleic acids are some of the potential candidates for response biomarkers. In addition, CYFRA 21-1 and CgA have shown responses to drug treatment in early studies.5
There are two types of response biomarkers: pharmacodynamic and surrogate endpoint biomarkers.
- Pharmacodynamic (PD)- These biomarkers convey information about a drug's effect on the human body. Combining this biomarker information with the patient's response to the drug can help design the next phases of clinical trials or aid in making an informed “go/no-go” decision regarding treatment. To fast-track PD biomarker development, National Institutes of Health’s (NIH) division of Cancer Treatment and Diagnosis (DCTD) has initiated a number of visionary programs, including the development of multiplex panel assays to detect possible drug targets of mTOR or ERK-MEK signaling pathway.16
- Surrogate- Surrogate biomarkers can help in clinical trial phase design if the primary biomarker for testing the efficacy of the treatment is unavailable. If disease progression cannot be directly measured via a standard biomarker, a surrogate marker assesses a drug's response to the given treatment. For example, for the HIV-AIDS pivotal clinical trial, CD4 measurement was used to identify disease progression instead of mortality rate, which would have resulted in an indefinitely long clinical trial.17,18
Biomarkers in Cancer Research
Cancer biomarkers have evolved in recent years, as researchers begin to understand the complexity of tumor biology. Because of the interactions among multiple proteins, there is a growing need to identify a panel of multiple molecules/biomarkers that would affect a drug treatment.19 For example, a panel of 70 marker genes is being tested in a clinical trial (MINDACT) that could convey information about disease relapse, which could aid in follow-up chemotherapy decisions.11,20 Precise biomarker identification can help in patient stratification and guide clinicians to make an informed decision about prospective treatment regimens. For example, mismatch repair (MMR) status, determined through analysis of 4 markers, can provide information on Lynch syndrome and appropriate patient treatment.
Across cancer biomarker testing assays, including tumor subtyping, somatic testing, genomic testing, and molecular testing/profiling, clinical validity and utility are challenging to achieve Therefore, evidence-based studies should demonstrate the clinical value of the tested cancer biomarker. Based on the evidence of the tumor biomarker, Hayes et al. have devised a scoring system to categorize a biomarker molecule.21 The scoring is based on the biomarker's status in the biological progression of the disease and the corresponding clinical manifestation (improvement in overall survival, better disease outcome, etc.), or antibody binding to the desired epitope.
Biomarkers in Spatial Research
Spatial biology has paved exciting avenues for improved patient care. Spatial transcriptomics, genomics, proteomics, and single-cell sequencing can all aid in identifying a specific biomarker in the context of the cellular microenvironment. Especially in the complex tumor microenvironment, biomarker detection and expression may change from one tissue to another. Hence, spatial research has now become an essential tool for precision medicine.22 Armed with spatially resolved "-omics" information, clinicians will have the opportunity to accurately predict the relevant biomarkers for a specific sample in situ, thereby significantly upgrading the personalized medicine field. The amalgamation of artificial intelligence, multiplexing, and digital pathology has the potential to change clinical outcomes radically.23 The advent of spatial biology has enabled the detection of more than 50 biomarkers simultaneously on a tissue sample to monitor disease progression.24,25,26 Several studies have shown that the presence of multiple T-cell antigens (CD8, CD3, PD-1/PD-L1, etc.) in the spatially resolved environment helps to predict the patient's progression-free survival better.27,28 Spatial biomarker detection is also helping in the management of neurological and oncogenic diseases (for example, Alzheimer's and head and neck cancer). To date, spatially resolved biomarkers have not been approved by the food and drug administration (FDA)22, but rapid strides have been taken to validate and verify the use of these biomarkers.
Discussion
Biomarkers are a valuable tool to inform clinicians about appropriate disease treatment. Identifying reproducible and validated biomarkers is necessary to produce consistent results for a specific disease type.29 Additionally, the determination of biomarker toxicity, clinical utility, and outcome are the factors that a stakeholder considers when deciding on a biomarker for disease management.30 Biomarkers have increased in prominence because of their ability to aid in stratifying patients according to the requisite disease management regimen. For example, biomarkers like PD-L1, ER, PR, HER2, and mismatch repair proteins have extensively aided in improving the patient survival rate in breast, lung, colorectal, and other types of cancers.31
There are many types of biomarkers, including predictive, prognostic, and diagnostic, and their categorization, biology, and applications are ever evolving. The FDA, in its guidelines, has recommended features of a biomarker that would allow its approval by the health authority and use in clinical practice; some of these include performing gap assessment, analyzing the risks and benefits, proposing a type of biomarker, and robust evidence backing the biomarker's performance supported by exhaustive statistical analyse. A schematic of the FDA’s recommendation on a biomarker is presented in Figure 1. Additionally, realizing the translational value of biomarkers, the health authority and the NIH have developed several resources, including BEST (Biomarkers, EndpointS, and other Tools)32, to clarify biomarker types and their potential use in medical research.
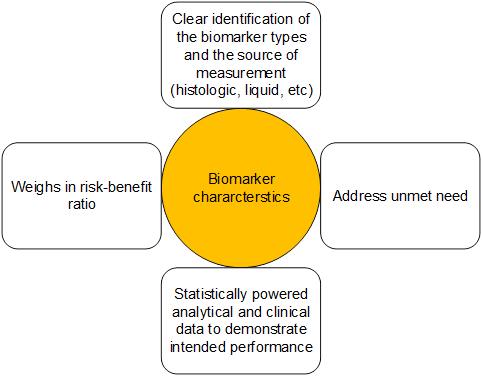
Spatial biology is now giving rise to new possibilities and techniques which would allow identification of a biomarker type in the context of tumor or cellular microenvironment. Proteomics, transcriptomics, and genomics are disruptive technologies that could revolutionize understanding of biomarkers which would enable clinicians to develop novel therapeutic strategies for improved patient outcomes.
Conclusion
There is enormous potential in the biomarker field. With a wealth of information provided by rapidly advancing new technologies, researchers are only now discovering the clinical potential of biomarkers and the promise they hold in solving clinical problems across many diseases.
About the presenters

Shubham Dayal is a Senior Medical Writer at Leica Biosystems and has over 10 years of experience in regulatory/preclinical/clinical writing for studies that are at different stages of the product lifecycle. Shubham has a PhD in Cell and Molecular Biology from the University of Toledo and a Master's in Regulatory Affairs from Northeastern University and has co-authored multiple peer-reviewed articles and poster presentations. He is an active member of the Regulatory Affairs Professional Society and American Medical Writers Association and holds certifications related to scientific writing. In his current role, Shubham's goal is to create awareness for our customers in ways that can advance scientific communication and ultimately improve patient outcomes.

Jack obtained his doctorate in molecular and cellular pathology and performed post-doctoral studies on cancer epigenetics and cardiovascular post-translational modifications. He has worked in externally facing roles at Leica Biosystems for 7 years, and currently works in partnership with leading pathologists and researchers to advance scientific study at the cutting edge of anatomic pathology research.
References
- Califf RM. Biomarker definitions and their applications. Exp Biol Med (Maywood). 2018;243(3):213-221. doi:10.1177/1535370217750088
- What are the most common biomarkers? | NanoString. Accessed December 27, 2023. https://nanostring.com/blog/most-common-biomarkers/
- Biomarkers. Accessed December 27, 2023. https://www.niehs.nih.gov/health/topics/science/biomarkers/index.cfm#:~:text=A%20biomarker%20(short%20for%20biological,warning%20systems%20for%20your%20health.
- Biomarkers In Risk Assessment: Validity And Validation (EHC 222, 2001). Accessed December 27, 2023. https://www.inchem.org/documents/ehc/ehc/ehc222.htm
- Amin S, Bathe OF. Response biomarkers: re-envisioning the approach to tailoring drug therapy for cancer. BMC Cancer. 2016;16(1):850. doi:10.1186/s12885-016-2886-9
- Susceptibility/Risk Biomarker - BEST (Biomarkers, EndpointS, and other Tools) Resource - NCBI Bookshelf. Accessed December 27, 2023. https://www.ncbi.nlm.nih.gov/books/NBK402288/
- 7 Types of Biomarkers. Accessed December 27, 2023. https://www.atlasantibodies.com/blog/7-types-of-biomarkers/
- Steffen P, Kwiatkowski M, Robertson WD, et al. Protein species as diagnostic markers. J Proteomics. 2016;134:5-18. doi:10.1016/j.jprot.2015.12.015
- Prognostic Biomarker - BEST (Biomarkers, EndpointS, and other Tools) Resource - NCBI Bookshelf. Accessed December 27, 2023. https://www.ncbi.nlm.nih.gov/books/NBK402289/
- Jin TY, Park KS, Nam SE, Yoo YB, Park WS, Yun IJ. BRCA1/2 serves as a biomarker for poor prognosis in breast carcinoma. Int J Mol Sci. 2022;23(7). doi:10.3390/ijms23073754
- Oldenhuis CNAM, Oosting SF, Gietema JA, de Vries EGE. Prognostic versus predictive value of biomarkers in oncology. Eur J Cancer. 2008;44(7):946-953. doi:10.1016/j.ejca.2008.03.006
- Bustoros M, Mouhieddine TH, Detappe A, Ghobrial IM. Established and novel prognostic biomarkers in multiple myeloma. Am Soc Clin Oncol Educ Book. 2017;37:548-560. doi:10.1200/EDBK_175175
- Kerr DJ, Yang L. Personalizing cancer medicine with prognostic markers. EBioMedicine. 2021;72:103577. doi:10.1016/j.ebiom.2021.103577
- Stickeler E. Prognostic and predictive markers for treatment decisions in early breast cancer. Breast Care (Basel). 2011;6(3):193-198. doi:10.1159/000329471
- Safety Biomarker - BEST (Biomarkers, EndpointS, and other Tools) Resource - NCBI Bookshelf. Accessed December 27, 2023. https://www.ncbi.nlm.nih.gov/books/NBK402287/
- Development | NExT Resources | NExT. Accessed December 27, 2023. https://next.cancer.gov/developmentresources/pd_biomarker.htm
- Katz R. Biomarkers and surrogate markers: an FDA perspective. NeuroRx. 2004;1(2):189-195. doi:10.1602/neurorx.1.2.189
- Peto T. Surrogate markers in HIV disease. J Antimicrob Chemother. 1996;37 Suppl B:161-170. doi:10.1093/jac/37.suppl_b.161
- Biomarker Testing for Cancer Treatment - NCI. Accessed December 27, 2023. https://www.cancer.gov/about-cancer/treatment/types/biomarker-testing-cancer-treatment#:~:text=Biomarker%20testing%20is%20a%20way,how%20certain%20cancer%20treatments%20work.
- Cardoso F, van’t Veer LJ, Bogaerts J, et al. 70-Gene Signature as an Aid to Treatment Decisions in Early-Stage Breast Cancer. N Engl J Med. 2016;375(8):717-729. doi:10.1056/NEJMoa1602253
- Hayes DF, Bast RC, Desch CE, et al. Tumor marker utility grading system: a framework to evaluate clinical utility of tumor markers. J Natl Cancer Inst. 1996;88(20):1456-1466. doi:10.1093/jnci/88.20.1456
- Biocompare: The Buyer’s Guide for Life Scientists. Accessed December 27, 2023. https://www.biocompare.com/Editorial-Articles/596937-Discovering-New-Biomarkers-Using-Spatial-Approaches/
- The New Frontier of Spatial Biomarkers: Digital Pathology and Spatial Genomics. Accessed December 27, 2023. https://globalforum.diaglobal.org/issue/august-2023/the-new-frontier-of-spatial-biomarkers-digital-pathology-and-spatial-genomics/
- From images to insights: deep spatial phenotyping reveals disease and treatment biomarkers. Accessed December 27, 2023. https://www.nature.com/articles/d44224-023-00025-5
- New method identifies spatial biomarkers of Alzheimer’s disease progression in animal model | Broad Institute. Accessed December 27, 2023. https://www.broadinstitute.org/news/new-method-identifies-spatial-biomarkers-alzheimer%E2%80%99s-disease-progression-animal-model
- Spatial Proteomics Maps Head and Neck Tumors, Targets Biomarkers. Accessed December 27, 2023. https://www.genengnews.com/topics/omics/spatial-proteomics-maps-head-and-neck-tumors-targets-biomarkers/
- Saberzadeh-Ardestani B, Graham RP, McMahon S, et al. Immune Marker Spatial Distribution and Clinical Outcome after PD-1 Blockade in Mismatch Repair-deficient, Advanced Colorectal Carcinomas. Clin Cancer Res. 2023;29(20):4268-4277. doi:10.1158/1078-0432.CCR-23-1109
- Hammerl D, Martens JWM, Timmermans M, et al. Spatial immunophenotypes predict response to anti-PD1 treatment and capture distinct paths of T cell evasion in triple negative breast cancer. Nat Commun. 2021;12(1):5668. doi:10.1038/s41467-021-25962-0
- Strimbu K, Tavel JA. What are biomarkers? Curr Opin HIV AIDS. 2010;5(6):463-466. doi:10.1097/COH.0b013e32833ed177
- Hayes DF. Defining clinical utility of tumor biomarker tests: A clinician’s viewpoint. J Clin Oncol. 2021;39(3):238-248. doi:10.1200/JCO.20.01572
- Triple-negative Breast Cancer | Details, Diagnosis, and Signs | American Cancer Society. Accessed December 27, 2023. https://www.cancer.org/cancer/types/breast-cancer/about/types-of-breast-cancer/triple-negative.html
- FDA-NIH Biomarker Working Group. BEST (Biomarkers, EndpointS, and Other Tools) Resource. Food and Drug Administration (US); 2016.
Leica Biosystems content is subject to the Leica Biosystems website terms of use, available at: Legal Notice. The content, including webinars, training presentations and related materials is intended to provide general information regarding particular subjects of interest to health care professionals and is not intended to be, and should not be construed as, medical, regulatory or legal advice. The views and opinions expressed in any third-party content reflect the personal views and opinions of the speaker(s)/author(s) and do not necessarily represent or reflect the views or opinions of Leica Biosystems, its employees or agents. Any links contained in the content which provides access to third party resources or content is provided for convenience only.
For the use of any product, the applicable product documentation, including information guides, inserts and operation manuals should be consulted.
Copyright © 2024 Leica Biosystems division of Leica Microsystems, Inc. and its Leica Biosystems affiliates. All rights reserved. LEICA and the Leica Logo are registered trademarks of Leica Microsystems IR GmbH.